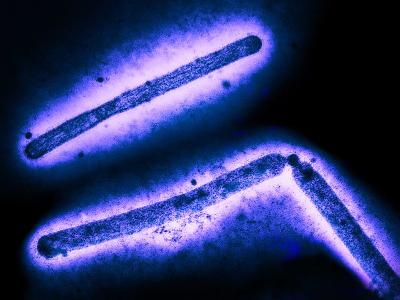
Mar 22, 2010 (CIDRAP News) The US Food and Drug Administration (FDA) today advised healthcare practitioners to temporarily stop using GlaxoSmithKline's (GSK's) Rotarix rotavirus vaccine while it investigates the presence of porcine circovirus 1 (PCV1) in the product, a finding that isn't currently thought to pose a safety risk.
In a press release today, the FDA said an American academic research team, using a novel technique, found DNA evidence of PCV1 in the vaccine. A health alert from the Centers for Disease Control and Prevention (CDC) e-mailed to clinicians today said GSK reported the academic group's finding to the FDA on Mar 15. The virus isn't known to cause disease in animals or humans.
Follow-up studies by GSK and the FDA found that viral components of the PCV1 virus have been in the vaccine since it was developed and throughout clinical trials. The CDC alert added that evidence of the virus was found in the cell bank and seed used to produce the Rotarix vaccine. However, the FDA emphasized that the vaccine has had an excellent safety record, before and after approval.
Rotarix is one of two FDA-approved rotavirus vaccines. Most US children have received the other rotavirus vaccine, RotaTeq, made by Merck. The FDA said no PCV1 components have been detected in the Merck vaccine, which the CDC said is made using a different process than the Rotarix vaccine.
The two vaccines are given by mouth to young infants to prevent rotavirus disease, which can cause severe diarrhea, dehydration, and death, particularly in infants in low- and middle-income countries.
CDC Director Tom Frieden, MD, said in the FDA release that the benefits of the Rotarix vaccine in many countries outweighs any risks, and he predicted that many will continue vaccinating children while scientists investigate the PCV1 findings.
The FDA said it would determine the full extent of the PCV1 findings and if it contains DNA fragments or intact virus. Within the next 4 to 6 weeks the FDA will convene an advisory group to make recommendations about use of rotavirus vaccines.
See also:
Mar 22 FDA press release