Sep 11, 2012 (CIDRAP News) – A Korean-US research team has identified an H1N2 strain of swine influenza capable of killing ferrets and spreading among them by respiratory droplets, underlining the continuing threat of swine flu to humans amid a wave of swine-origin flu cases in Americans exposed to pigs at agricultural fairs.
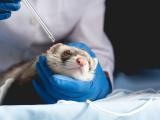
The researchers exposed groups of ferrets, which are considered the best animal models for human flu, to four swine flu viruses isolated in Korea—two H1N2 and two H3N2. One of the H1N2 isolates caused severe or fatal illness in the animals and spread to other ferrets housed nearby without direct contact, according to the scientists' report in the Proceedings of the National Academy of Sciences.
In addition, when the scientists analyzed the virus in the ferrets that were infected secondarily, they found that it had two mutations and was able to spread even more readily to other ferrets.
The findings come 3 years after another swine-origin flu virus, the 2009 H1N1 strain, triggered a pandemic, which the authors say marked the broadest known spread of a swine-origin strain in humans.
Richard J. Webby, PhD, a co-author of the study, said the newly identified virus, dubbed Sw/1204, is closely related to swine H1N2 viruses in the United States. Experts say the H1N2 subtype is fairly common in swine, and a few swine-origin H1N2 infections, called H1N2v, have been identified in humans in recent years, including four cases in people who were exposed to pigs at the Minnesota State Fair in August.
Another swine-origin virus, variant H3N2, or H3N2v, has infected nearly 300 people in the United States this summer, most of whom showed pigs or were otherwise exposed to pigs at county and state fairs. Sixteen patients were hospitalized, and one person, a woman who had preexisting health problems, died.
Webby, who is with St. Jude Children's Research Hospital in Memphis, said Sw/1204 probably represents only a minor threat to humans, because it is related to the 2009 H1N1 strain, and therefore anyone who was exposed to the latter would have "considerable immunity" to it.
Still, the findings show the continuing pandemic potential of reassortant swine flu viruses, the researchers say.
The authors write that, because swine flu surveillance is "abysmal," it is unclear whether swine flu subtypes other than H1N1 could spread among humans and cause serious illness. Their aim was to assess the zoonotic potential of representative Korean "triple-reassortant" H1N2 and H3N2 viruses that are related to North American strains. "Triple-reassortant" generally refers to flu viruses that contain genes from avian, human, and swine flu strains.
They picked four isolates: two H1N2 viruses collected in 2009 (before the pandemic) and two H3N2 collected identified in 2007. Groups of three ferrets were exposed to high doses of these strains via the nose and throat and then were observed for 14 days.
Three of the strains grew only moderately, were undetectable in upper respiratory samples after a week, and caused no clear signs of disease, according to the report.
But the ferrets dosed with the Sw/1204 (H1N2) virus got very sick starting 3 days after infection, with major weight loss, labored breathing, high fever, and other signs. One of the animals died after 7 days, and the other two were euthanized (at 7 and 10 days) because of their severe illness.
To test transmissibility, the authors placed uninfected ferrets in cages near each of the infected ones, but not close enough for direct contact. Only Sw/1204 spread via respiratory droplets to all three uninfected ferrets. One of these contact ferrets died 12 days after exposure, and another was euthanized because of severe illness.
A comparative molecular analysis revealed that the virus from the contact ferrets, called CT-Sw/1204, had acquired two mutations—one each in its hemagglutinin (HA) and neuraminidase (NA) proteins—that distinguished it from the original or wild-type Sw/1204 virus. Evidence indicated that these mutations, called HA225G and NA315N, evolved mainly during viral replication in the originally inoculated animals.
In further experiments, the researchers inoculated additional groups of ferrets with CT-Sw/1204 and found that it proved lethal a day earlier and spread more quickly to other ferrets (as soon as 1 day after exposure, versus 4 to 5 days) than did the original virus.
In still other experiments, the authors found that the CT-Sw/1204 grew more efficiently than the original Sw/1204 in human respiratory cell cultures. In freshly biopsied lung tissue samples, for example, the original virus showed minimal infectivity, whereas CT-Sw/1204 grew to high titers within 36 hours—a finding consistent with the ferret results.
To examine the effects of the two mutations on viral pathogenicity and transmissibility, the authors also used reverse genetics techniques to generate Sw/1204 viruses with just one of the mutations, and then exposed mice and ferrets to these recombinant strains. The results suggested that both of the changes may have contributed to virulence and transmissibility.
The team also determined that the recombinant strains of Sw/1204, with or without the two mutations, caused only mild illness in pigs and were susceptible to neuraminidase inhibitors used to treat flu.
"Overall, we have presented evidence proposing the role of HA225G and NA315N as potential virulence markers of TRSw [triple-reassortant swine] influenza viruses in mammalian hosts," the report states. "Our findings further support the continuing pandemic potential of TRSw influenza viruses circulating in pigs," thereby showing that surveillance in pigs is "vital and beneficial."
Webby said it is very unusual for a swine flu virus to cause severe illness in ferrets and that swine flu viruses typically don't spread easily among ferrets, either. By comparison, ferret experiments with the 2009 H1N1 virus showed that it generally was not lethal, though it did cause a few cases of severe illness, he told CIDRAP News.
"I'd say the generalization is that the pandemic virus was a bit more able to cause disease in most [animal] models than the average swine virus," he said.
Concerning the potential risk of Sw/1204 to humans, Webby said, "I think this particular one, the threat is fairly minor because it's a not-so-distant cousin of the pandemic virus itself, so anyone who was exposed to the pandemic virus would have considerable immunity to this particular strain."
Using serologic tests (hemagglunination inhibition), Webby and his colleagues found that antibodies to the 2009 H1N1 virus cross-reacted to a considerable degree with Sw/1204, suggesting that previous exposure to 2009 H1N1 or vaccination against it is probably beneficial, their report says. But it cautions that this remains to be confirmed.
Webby said the H1N2 viruses used in the study are "quite closely related" to swine H1N2 strains in the United States. "The viruses in Korea were likely imported from the US," he said. "We've done some risk assessments on some viruses circulating in the US and haven't seen any phenotypes with the properties of this [Sw/1204] virus."
Although the study generated a mutant H1N2 virus that spread more easily among ferrets while maintaining its virulence, Webby said the study did not violate the voluntary moratorium that leading flu researchers announced in January on studies involving the generation of H5N1 avian flu viruses with increased transmissibility in mammals.
The moratorium was declared by 39 leading researchers, including Webby, in response to the controversy over proposed publication of two studies in which scientists generated H5N1 viruses that could spread in ferrets by the aerosol route. Out of concern about the potential for release of a dangerous virus, a US committee of experts initially recommended that full versions of the studies not be published, but the group later reversed itself, and the studies were published in May and June.
Webby noted that the moratorium specifically targeted research on highly pathogenic H5N1 viruses, with no mention of other subtypes. Also, much of the research in the new report was done before the moratorium was launched.
He added, "A critical piece of the moratorium was that we shouldn't stop characterizing viruses that are out there. Essentially this was a characterization of a virus that was isolated from pigs; it wasn't designed to create a more virulent and more transmissible virus.
"There were some mutations that occurred during the analysis of this virus, and they were described in this paper. Certainly no mutations were intentionally introduced that enhanced transmission or pathogenicity."
Webby also commented that swine flu viruses are not on the list of 15 agents and toxins cited in the US government's policy for oversight of "life sciences dual-use agents of concern," which was announced in March. It calls on federal agencies to review the potential risks of federally funded studies involving those agents and toxins.
The research moratorium was originally intended to last just 60 days, but it has been extended indefinitely as scientists await further development of guidelines for the review of dual-use studies.
In the meantime, Webby said, "We're all thinking hard about what experiments we do, the potential implications. The actual review processes that need to be done will be coming down the pipeline."
Pascua PNQ, Song MS, Lee JH, et al. Virulence and transmissibility of H1N2 influenza virus in ferrets imply the continuing threat of triple-reassortant swine viruses. Proc Natl Acad Sci 2012 Sep 10 (early online release) [Abstract]
See also:
Flu researchers' Jan 20 moratorium statement
US policy for oversight of dual-use research