Oct 26, 2012 (CIDRAP News) – The compounding pharmacy that produced tainted steroid injections implicated in a multistate fungal meningitis outbreak had problems maintaining the clean room where the drugs were processed, according to a federal report today that found, for example, greenish residue on the ceiling.
Inspectors from the US Food and Drug Administration (FDA) were at New England Compounding Center (NECC) in Framingham, Mass., in early October, and they detailed their initial observations regarding possible violations of federal or other regulations in a 483 report that appears on the FDA's Web site.
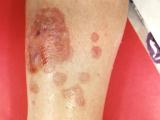
Meanwhile, the US Centers for Disease Control and Prevention (CDC) said 10 more fungal meningitis cases have been reported in the outbreak, including one more death, according to an update today. The new cases raise the outbreak total to 338 and 25 deaths.
The recalled steroids were also used to treat joint problems, and two more peripheral joint infections linked outbreak were reported, pushing that number to seven. The number of affected states remained at 18.
Residue findings
In a press release today, the FDA said that producing sterile drugs in a properly functioning and maintained clean room reduces the risk of introducing microbial contamination into the drug during processing, which includes the final filling step.
When FDA officials examined 321 vials of methylprednisolone acetate from one of the three recalled lots, they found greenish-black foreign matter in 83 of them, and white filamentous material in 17, according to the report.
The report said NECC's sterility test on one 5 milliliter vial of bulk methylprednisolone acetate from the recalled lot found no contamination, but the FDA's own tests on vials from the same lot found evidence of microbial growth in all 50 vials tested, with microscopic evidence of fungus in one of them.
A review of the raw materials used to make the recalled steroid injections found no indications that they were sterile or that the steam autoclave cycle NECC used to sterilize the steroid suspensions was effective.
Inspectors reviewed NECC's environmental monitoring records from January to September for two clean rooms and found that the firm did not investigate bacterial and mold levels that exceeded their action limits. Also, officials didn't find any indication that the company took any corrective actions to remove the contaminants.
The report described problems with other autoclaves, such as greenish-yellow discoloration on the lens inside a machine used in the final sterilization step.
Heating, cooling system issues
When they examined the heating and air conditioning system, inspectors found that the rooftop unit was located about 100 feet from a recycling facility that handled mattresses and plastics, and large equipment such as freight trucks that produced airborne particulates were observed at the facility. They also found dark particulate and white filamentous substances covering the louvers of a heat and air conditioning return located behind one of the autoclaves.
A leaky boiler near one of the workrooms produced puddles and wet surfaces where officials observed thick white debris and thick black granular material. The FDA report said that inspectors also found that a tacky mat between the prep room and warehouse was brown and soiled.
In its press release, the FDA said officials are still working on an establishment inspection report. It said the FDA will use that report, the 483 report, responses from the company, and any other relevant information it receives when deciding if further action is appropriate.
The agency said it continues to work closely with the CDC and state partners on the outbreak investigation.
Health officials question oversight
In related news, officials from the Maryland Department of Health and Mental Hygiene said in a commentary published today in the Journal of the American Medical Association (JAMA) that only about 2% of the estimated 7,500 US compounding centers participate in voluntary accrediting and that 1% to 3% of all prescriptions are compounded.
They wrote that state pharmacy boards provide inconsistent oversight and often lack resources to police these centers. The officials added that the FDA, in spite of judicial and political efforts to thwart oversight, has issued consumer advisories about compounding centers and sought to retain authority over pharmacies that function as large-scale manufacturers.
"In the case of the New England Compounding Center, however," they wrote, "oversight efforts failed to prevent harm."
They concluded, "The debacle at the New England Compounding Center, however, indicates that compounding has yet to fully emerge from the Dark Ages. Congressional hearings will call attention to the need to stop certain high-risk compounding practices and more closely oversee those that remain.
"Legislation clarifying FDA's authority and empowering the agency to establish national standards for compounding, including adverse event reporting, should follow."
Editorial Director Jim Wappes contributed to this story.
See also:
Oct 26 FDA 483 inspection report
Oct 26 CDC fungal meningitis update
Oct 26 FDA press release
Oct 26 JAMA commentary