Oct 11, 2012 – Federal officials today announced that testing in the multistate meningitis outbreak has found fungus in 50 unopened vials of the steroid linked to the outbreak and that Michigan officials reported the first joint infection in a patient who received the recalled drug.
Meanwhile, the number of patients infected in the outbreak climbed to 170, with 33 cases added to the total since yesterday, according to the Centers for Disease Control and Prevention (CDC) today. Two more deaths were reported, raising the total to 14, and Idaho reported its first infection, making it the 11th state to be affected by the outbreak.
At a media briefing today, Deborah Autor, JD, deputy director of Global Regulatory Operations and Policy with the US Food and Drug Administration (FDA) expressed condolences to the patients and families affected by the outbreak. She said FDA investigators are working around the clock alongside the CDC to identify the source of the contamination and flesh out the problems that led to it.
Autor said fungus was found in unopened vials from both the New England Compounding Center (NECC) facility that produced the methylprednisolone acetate injections as well as some of vials retrieved from clinics that received them.
Tests are under way to identify the types of fungus that are in the vials, Autor said. One of the earliest patients sickened in the outbreak was infected with Aspergillus fumigatus, but since then most of the organisms identified have been Exserohilum, a black mold that health officials say has never been known to cause meningitis before.
FDA and officials from Massachusetts are jointly inspecting the NECC facility and are looking into the practices of other compounding pharmacies that have business ties with the company, she said.
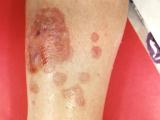
J. Todd Weber, MD, incident manager of the CDC's fungal meningitis outbreak investigation, told reporters that the joint infection that officials suspect is related to the outbreak was identified in a Michigan patient whose ankle became infected after receiving a joint injection with a steroid from recalled lots. He said tests are under way to determine if the source of the infection is fungal.
CDC officials have said the steroids were also used for joint-space injections for knee, shoulder, and ankle problems, and they have warned that they expect to receive reports of fungal joint infections in addition to fungal meningitis in patients who received epidural injections for back pain. He said the CDC experts and their panel of fungal disease experts are working on treatment guidance for fungal joint infections.
Weber credited states and their public health labs for speeding the outbreak investigation. He said Tennessee officials quickly identified the first outbreak cases, Virginia's lab was the first to identify Exserohilum in the samples, and Michigan's vigilance led to identification of the joint infection.
So far the CDC's emergency operations center and its partners have successfully contacted about 12,000 of the 13,000 patients who were exposed to the recalled injections, Weber said.
The involvement of Exserohilum fungus complicates identification and treatment of infections, because it can be difficult to detect on tests. "This is new territory for public health," Weber said.
Patients linked to the outbreak who have meningitis symptoms should still received the recommended powerful two-drug treatment, even if tests are negative, he said, urging clinicians to check the CDC's Web site frequently, since recommended drugs, dosages, and durations could change based on new findings.
In other developments, the FDA's Autor and Madeleine Biondolillo, MD, who directs the Massachusetts Department of Public Health's Bureau of Health Care Safety and Quality, said NECC appears to have violated its operating license, which stipulates that drug doses must be prepared for patient-specific prescriptions. The recall involved three lots of the steroid preparation, which were sent to 23 states and used to inject about 13,000 patients, according to CDC estimates.
Autor said one of the goals of the investigation is to identify regulatory gaps that apply to compounding pharmacies. She acknowledged that NECC received a warning from the FDA that the company's practices resembled those of a pharmaceutical company's, but she said the agency has limits on its authority to do a detailed follow-up, such as looking at the company's distribution records.
She said the practice of the pharmacy is changing and that it's a shame that it takes a crisis to put risk-based controls in place.
See also:
Oct 11 CDC outbreak update
Oct 10 CIDRAP News story "Meningitis outbreak poses questions for experts, clinicians"