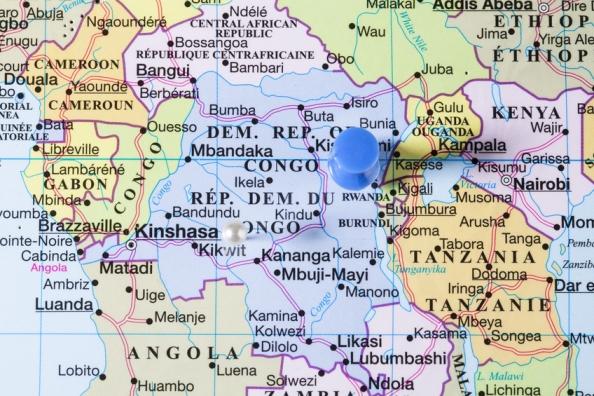
Yesterday the World Health Organization (WHO) released more details of its plan to launch an emergency yellow fever vaccination campaign in Angola and the Democratic Republic of Congo (DRC). The agency earlier yesterday shared its updated response plan to address the outbreak in east Africa.
The organization said it will begin the pre-emptive vaccination campaign in July. Phase one will focus on the high-traffic, high-trade corridor between Angola and the DRC, targeting 2.3 million people in Angola and 2.9 million in the DRC. In addition to the 45- to 60-mile-long corridor, the campaign will target 9.7 million peopling living in Kinshasa.
Currently, the WHO and its partners are offering vaccinations at major points of entry in both countries: Luanda in Angola and Kinshasa, Lubumbashi, and Matadi in the DRC.
Overcoming vaccine shortages
The WHO said that to date it has delivered 15 million doses of the vaccine to Angola and the DRC through its International Coordinating Group. But a worldwide shortage in yellow fever vaccine looms large in the WHO's plan to increase its vaccination efforts in the urban areas most at risk for an outbreak. As of Jun 16, the total number of vaccines in stock is about 4 million, with another 13 million expected by the end of July.
"While WHO is working with partners and vaccine manufacturers to increase vaccine production and replenish the emergency stockpile currently being used for this outbreak, it is vital to interrupt transmission, especially in cross-border areas to rapidly bring this outbreak under control and halt further international spread," said Matshidiso Moeti, MD, WHO regional director for Africa.
The vaccine is affordable; the proposed campaign would cost $2 per dose ($1 for the vaccine and $1 for operational costs). The WHO reports that the Angolan government is committed to funding half the vaccination costs; the DRC is eligible to receive funds from Gavi, the Vaccine Alliance.
Last week, the WHO said it was considering diluted dosing of the vaccine to stretch supplies, stating this method would be suitable for urban populations. A dose one fifth the strength of the standard vaccine would provide immunity from yellow fever for 12 months. A full dose offers lifelong protection.
In case of a vaccine shortage, the WHO stated it would target age-groups that have the highest illness rates (6 months to 45 years). WHO previously stated that its goal was to immunize over 80% of the population in affected districts.
Yellow fever vaccine is one of the safest and most effective vaccinations offered. In statements made to the World Health Assembly in May, WHO Director-General Margaret Chan, MD, MPH, said, "The world failed to use an excellent preventive tool to its full strategic advantage" by allowing yellow fever vaccine campaigns to wane in the last 40 years.
Chan said rapid urbanization and changes in land-use patterns made Angola a perfect storm for the yellow fever outbreak that began in January in Luanda, its capital.
Cases rise in Angola, DRC
Meanwhile, the WHO's yellow fever situation report today said there are now 3,294 suspected cases in Angola, 157 more than last week. Two deaths were reported in the country this week, making the total number of reported deaths 347 (115 lab-confirmed). The outbreak continues to hit cities Luanda and Huambo the hardest, with 1,833 and 543 suspected cases, respectively.
The DRC reported 62 more suspected cases this week, raising its count to 1,044. In addition, 75 deaths have been reported. Of the 68 lab-confirmed cases of yellow fever in the DRC, 59 were imported from Angola.
Kenya and China remain the only two counties outside of the outbreak zone reporting confirmed yellow fever cases imported from Angola.
See also:
Jun 22 WHO news release
Jun 23 WHO situation report
Jun 22 CIDRAP News story "WHO details $72 million yellow fever response plan"