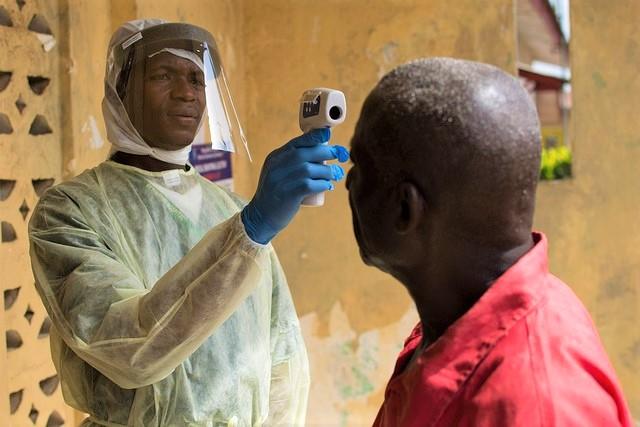
Over the past 4 days, the Democratic Republic of Congo (DRC) reported three more confirmed Ebola cases, two from Beni and one from Butembo, the second case to be detected in the large urban area.
World Health Organization (WHO) officials have said Beni, where community resistance has been reported in some areas, is an emerging hot spot in the DRC's latest outbreak, and the newly identified case from Butembo comes less than a week after the first case was found in the city, a commercial and transportation hub that has a population of about 1 million people.
Butembo patient fled from Ituri
The two new cases in Beni were noted in the health ministry's report for Sep 8, which also noted another death in an earlier confirmed Ebola patient from Beni.
In today's report, the health ministry said the newly confirmed case in Butembo involves a man who got sick in August and was admitted to a health center in Ituri province. He refused transfer to the Mangina Ebola treatment center, however, and fled to Butembo, where his illness progressed.
His blood samples were negative for the virus, but his sperm sample was positive.
During his recovery, his wife visited him, later got sick, and was confirmed as positive for Ebola. Her illnesses is among the confirmed cases from Mandima in Ituri province.
Also in today's report, the health ministry reported another death in an earlier confirmed case form Beni.
The new developments lift the overall outbreak total to 132 cases, including 101 confirmed and 31 probable. So far, 91 deaths have been reported, and health officials are still investigating 9 suspected cases.
Vaccination teams continue to make steady progress with administering the VSV-EBOV vaccine, and 7,780 people have now been immunized.
Experts reassess investigational drugs
A group of WHO experts met on Aug 27 for the second time to update the ethical framework for assessing investigational drugs that could be used in the DRC's Ebola outbreak, and the WHO today released notes covering the consultation, which covered new information that's become available since the experts' first meeting.
In May, the group first assessed five potential treatments that were being considered for an DRC's outbreak in Equateur province that was ongoing at the time.
The report covers the same five drugs: Zmapp, remdesivir, REGN3470-3471-3479, favipiravir, and mAB114. Efforts are under way to develop protocols to rapidly establish randomized controlled trials, which the experts say are the best way to identify therapies that help Ebola patients.
The highest quality data support the use of ZMapp, and benefits outweigh the risks, the group notes. Data also supports the use of remdesivir, an antiviral drug, but clinical trials are needed to weigh the risks and benefits.
For REGN3470-3471-3479, a monoclonal antibody cocktail, data are promising and support the drug's use as another possible option the scientists note.
Though the monoclonal antibody mAb114 is already being used in the current DRC outbreak, concerted efforts are needed to study its risks and benefits, the experts concluded. So far, a few patients have received the drug in the DRC's outbreak and no toxicities have been reported in Ebola patients or clinical study subjects.
It's not clear if favipiravir is helpful, but it's important to do clinical trials to assess if it's beneficial REGN3470-3471-3479 or mAb114 aren't available, the experts said.
The team also discussed the use of combined investigational therapies and learned that a non-human primate study is planned to test the combinations.
See also:
Sep 8 DRC health ministry update
Sep 10 DRC health ministry update
Sep 10 WHO treatment page