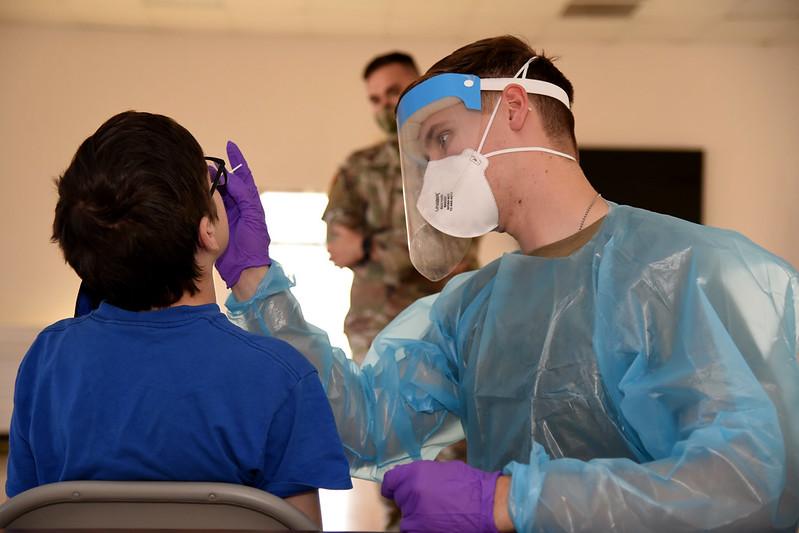
The United States surpassed 8 million confirmed COVID-19 cases today since the beginning of the pandemic, as a new surge of infections in the Midwest and West brings daily case numbers back to levels not seen since the peak days of July and early August.
The US reported 63,610 new COVID-19 cases yesterday, with 820 new deaths, according to the Johns Hopkins COVID-19 dashboard. It's the first time since Aug 1 that the nation has recorded more than 60,000 new infections in a single day. The overall number of new daily infections has been climbing since mid-September, and the national 7-day moving average of new cases is at 53,400.
The impact of the rise in new cases is reflected in the country's hospitalization numbers. Data from the COVID Tracking Project show that 37,308 people are currently hospitalized with COVID-19, up from 28,608 on Sep 28. Throughout the pandemic, COVID-19 hospitalizations have typically started rising a few weeks after increases in new infections.
Among the states where hospitals are starting to feel the strain is Wisconsin, where COVID-19 hospitalizations yesterday hit an all-time high of 1,043, with 264 people in intensive care units (ICUs), according to the Milwaukee Journal Sentinel. State health officials told the paper that ICUs across the state are more than 90% full, and hospitals in every region are reporting current or imminent staffing shortages.
Elsewhere in the country, Indiana health officials say COVID-19 hospitalizations have risen to their highest level since May, and the Kansas City Star reports that some Kansas City area hospitals are turning away ambulances with COVID-19 patients because their beds are already filled. Both Missouri and Kansas are seeing surges in COVID-19 cases and hospitalizations.
"I've got to ask people to continue to do, or start doing, what we know it takes to mitigate the spread of this disease," Kansas Gov. Laura Kelly said at a press conference earlier this week.
Pfizer says it won't seek vaccine authorization until November
Meanwhile, news today from drug maker Pfizer indicates that emergency authorization of a COVID-19 vaccine prior to election day—an idea that has been pushed repeatedly by President Donald Trump—is unlikely.
In a letter posted on the company's website, Pfizer Chairman and CEO Albert Bourla, DVM, said that, because the Food and Drug Administration (FDA) is requiring companies to provide 2 months of vaccine safety data on half of the phase 3 trial participants following final dose of the vaccine, the earliest the company could apply for emergency use authorization (EUA) is the third week of November.
Bourla, who has said in recent months that Pfizer will know by the end of October whether the vaccine is effective, said effectiveness would satisfy only one of the requirements of an EUA application. In addition to safety data, the company also has to show manufacturing data that demonstrate the quality and consistency of the vaccine that will be produced.
"So let me be clear, assuming positive data, Pfizer will apply for Emergency Use Authorization in the U.S. soon after the safety milestone is achieved in the third week of November," Bourla said.
The vaccine from Pfizer and German biopharmaceutical company BioNTech is one of several in phase 3 trials, and has widely been viewed as having the potential to be the first COVID-19 vaccine to be granted an EUA or approved. The late-stage trials of vaccine candidates from AstraZeneca and Johnson & Johnson are currently on hold due to adverse events among trial participants.
Although the FDA requirement that vaccine makers provide a median 2 months of follow-up data after completion of the full vaccine regimen received pushback from the White House, in early October FDA officials went ahead and included the requirement in guidance published on the FDA website. In an editorial published today in the New England Journal of Medicine, FDA officials defended their decision to require 2 months of data.
"From a safety perspective, a 2-month median follow-up (meaning that at least half of vaccine recipients in clinical trials have at least 2 months of follow-up) after completion of the full vaccination regimen will allow identification of potential adverse events that were not apparent in the immediate postvaccination period and will also provide greater confidence in their absence, if none are observed," Philip Krause, MD, and Marion Gruber, PhD, of the FDA's Office of Vaccine Research and Review, wrote.
Krause and Gruber said the 2 months of follow-up data could also provide more information on the duration of protection offered by a COVID-19 vaccine.
"Although 2 months of follow-up is insufficient to fully evaluate the duration of vaccine protection, substantial waning of protective responses might start to become apparent in the second month," they wrote. "Thus, a median of 2 months is the shortest follow-up period required to achieve some confidence that any protection against Covid-19 is likely to be more than very short-lived."
While the country awaits news on a potential COVID-19 vaccine, state health officials are racing to submit plans to the federal government on how'll they'll distribute a vaccine once it's approved, National Public Radio reports. The deadline for the first draft of those plans is today.
Also today, the Trump administration announced a deal with pharmacy chains CVS and Walgreens to administer a future COVID-19 vaccine to seniors and staff in nursing homes free of charge, the Associated Press reported. Under the program, trained staff from CVS and Walgreens would deliver and administer the shots.
COVID-19 racial disparities persist
In other US developments:
- Data released today by the Centers for Disease Control and Prevention in its Morbidity and Mortality Weekly Report show that US blacks and Hispanics continue to be disproportionately represented in COVID-19–associated deaths. Analysis of 114,411 deaths recorded from May through August in 50 states and the district of Columbia found that 51.3% were among non-Hispanic whites, 24.2% were Hispanic or Latino, and 18.7% were non-Hispanic black. Hispanics represent 18.5% of the US population, while non-Hispanic blacks represent 12.5%.
- Treasury Secretary Steven Mnuchin agreed to House Democrats' demands to include a national coronavirus-testing strategy in a broader coronavirus relief package, according to the Wall Street Journal. But the aid package, which is approaching $2 trillion dollars, still faces opposition from Senate Republicans, many of whom think it's too expensive.
- Former New Jersey Gov. Chris Christie, who was recently released from the hospital after spending 7 days in the ICU with COVID-19, said in a statement yesterday that Americans should take the coronavirus very seriously. "No one should be happy to get the virus and no one should be cavalier about being infected or infecting others," he said. Christie also said he regretted not wearing a mask to the Sep 26 Rose Garden ceremony for Judge Amy Coney Barrett or several debate prep sessions with President Trump.