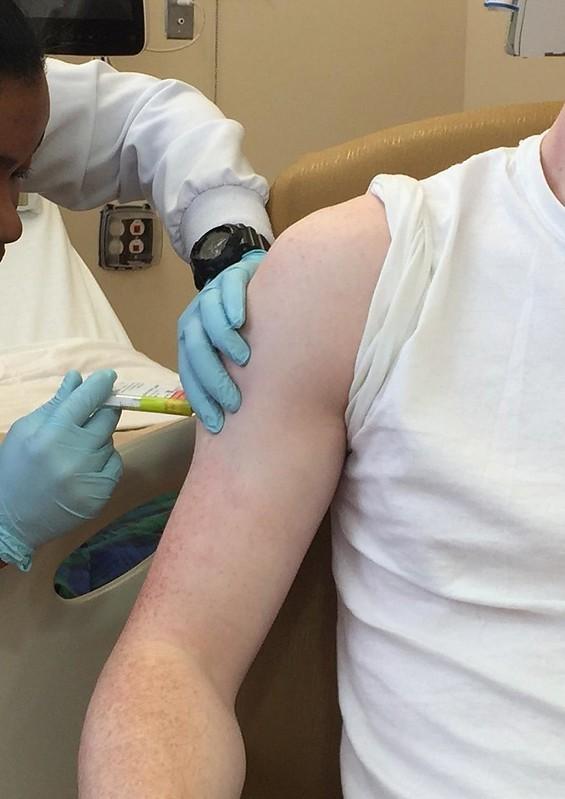
Development of vaccines against COVID-19 hinges on "unprecedented" and transparent cooperation among industry, government, and academia, according to a commentary by Anthony Fauci, MD, and Francis Collins, MD, PhD, of the National Institutes of Health (NIH), and two other US vaccine experts published yesterday in Science.
Noting that all vaccine platforms have advantages and disadvantages and underscoring the need for speed and flexibility of manufacture, safety, long-term efficacy, scale, affordability, vaccine stability, and a temperature-controlled supply chain, they said that "no single vaccine or vaccine platform alone is likely to meet the global need, and so a strategic approach to the multi-pronged endeavor is absolutely critical."
Public-private partnership, other steps
In particular, the authors highlight the NIH's ACTIV (Accelerating COVID-19 Therapeutic Interventions and Vaccines) private-public partnership that aims to conduct harmonized, randomized, controlled studies on several candidate vaccines in parallel to accelerate vaccine development, manufacture, and distribution.
To achieve the primary end points of development of antibodies protective against future COVID-19 infections and prevention of severe disease requiring hospitalization, vaccine trials must be conducted in a wide range of large epidemiologic and medical settings in the young, old, and underserved minority populations and include long-term follow-up, they said.
Controlled human challenge studies in which young volunteers are vaccinated and then exposed to COVID-19 are risky, may have limited utility, and would not predict similar effectiveness in older adults with underlying illnesses or prove reduction of spread to vulnerable populations, they said.
The authors also called for immediate funding of the global vaccine manufacturing infrastructure to meet the anticipated demand for hundreds of millions to billions of vaccine doses.
Given that major virus mutations have not been extensive so far, the authors expressed "cautious optimism that vaccines designed now will be effective against circulating strains 6 to 12 months in the future."
Latest vaccine developments
Several vaccines are in different stages of development. Today, Moderna announced that it has received a US Food and Drug Administration Fast Track designation for review of its mRNA-based COVID-19 vaccine. Fast Track facilitates the development and speeds review of therapies and vaccines for serious diseases that meet a medical need.
Moderna said that a phase 2 trial of the vaccine is expected to commence soon and that it is finalizing a protocol for a phase 3 trial to begin in early summer.
Novavax announced yesterday that it has received up to $384 million more in CEPI (Coalition for Epidemic Preparedness Innovations) funding for its adjuvanted COVID-19 vaccine, on top of the $4 million it received in March.
The company said that the money will advance clinical development of the vaccine, support rapid scale-up and large-scale manufacturing of the vaccine antigen (antibody-producing substance) and adjuvant, which boosts the body's immune response.