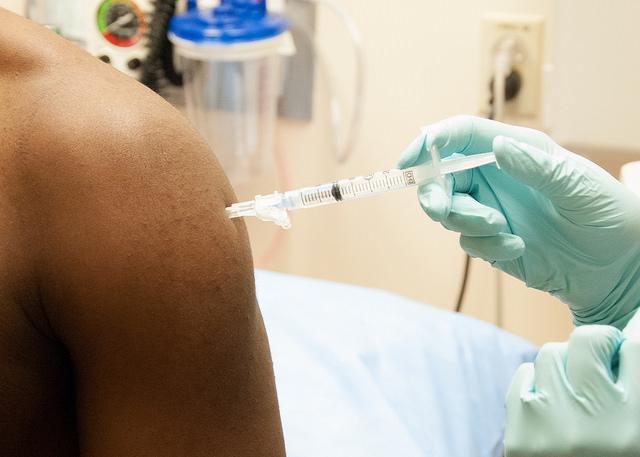
In a move designed to help a promising Ebola vaccine complete clinical trials and reach scaled-up manufacturing, the small biotech company NewLink Genetics and the pharmaceutical giant Merck today announced final details of an agreement that taps into the larger company's capacity and expertise.
In 2010 NewLink obtained the marketing rights for the experimental vaccine developed by the Public Health Agency of Canada (PHAC), one of the two experimental vaccines furthest along in testing. However, experts have aired concerns about whether the company has the resources to get the vaccine through testing, regulatory, and production steps to a point where it could be deployed in West Africa's Ebola outbreak. The vaccine, called VSV-EBOV, uses an Ebola virus protein spliced into a vesicular stomatitis virus (VSV).
Representatives of the companies met with Canadian health officials in Ottawa recently. Researchers who developed the vaccine have aired frustrations over perceived delays in testing and manufacturing, and some critics had earlier urged Canada to withdraw from its agreement with NewLink to clear the way for a larger company to move the product forward.
Agreement pays NewLink $30 million
NewLink had denied delays and said in late October that a batch of vaccine was almost ready and that it had contracted with European manufacturers to make more, according to previous reports.
Under the agreement, Merck will be granted the exclusive rights to the VSV-EBOV vaccine candidate and any follow-on products, according to press releases today from NewLink and Merck. The vaccine is under an exclusive licensing arrangement with BioProtection Systems, a wholly owned subsidiary of NewLink, and the PHAC retains noncommercial rights.
NewLink paid $205,000 for the license to VSV-EBOV vaccine and will get $30 million up front, with $20 million more when the vaccine enters efficacy testing, plus royalties if the vaccine is approved and cleared for marketing, the Canadian Press reported today. However, it won't receive royalties on doses sold to African countries and other low-income nations. The report also said Merck is well versed in making live-virus vaccines grown in vero cells, the same production substrate used to make VSV-EBOV.
Julie Gerberding, MD, MPH, president of Merck Vaccines, said in a statement, "Merck is committed to applying our vaccine expertise to address important global health needs and, through our collaboration with NewLink, we hope to advance the public health response to this urgent international health priority."
Charles Link, MD, NewLink's chairman and chief executive officer, said in the statement that Merck's development expertise, commercial leadership, and history of strategic alliances make the company an ideal partner to streamline the development of VSV-EBOV, and if effective, to make it available to people and communities at risk for Ebola infection.
The Canadian-developed vaccine is one of two that are furthest along in clinical trials. The other, called ChAD3, uses a modified chimpanzee adenovirus and was developed by the US National Institute of Allergy and Infectious Diseases (NIAID) at the National Institutes of Health (NIH) and GSK. Research studies have suggested that the Canadian vaccine would require only one dose, compared to two doses that may be needed for the NIH-GSK vaccine.
Phase 1 trials of VSV-EBOV are under way at the Walter Reed Army Institute of Research and at the NIH Clinical Center, with more under way or set to begin at research centers in Switzerland, Germany, Kenya, and Gabon in a World Health Organization (WHO)-coordinated effort and in Canada by the Canadian Immunization Research Network.
According to earlier reports, plans were in the works to skip phase 2 trials and launch phase 3 trials in Liberia and Sierra Leone sometime in early 2015.
Mali confirms another Ebola case
In other news, Mali's health ministry on Nov 22 confirmed a new Ebola infection and said testing is underway in two suspected cases, the Associated Press (AP) reported. The government released a statement that the patient who tested positive was placed in an isolation center for intensive treatment, but gave no other details.
Mali had an isolated imported case in the western part of the country, followed by another imported case that triggered a cluster of infections linked to a clinic cluster in Bamako, the capital. Based on the WHO case count for Mali as of Nov 21, confirmation of the new case would presumably push Mali's overall total to 7 cases, including 6 deaths.
The spread of the virus to Mali, a worrisome development, prompted a visit from WHO Director-General Margaret Chan, MD, MPH, over the weekend. The WHO said Chan met with Malian President Ibrahim Boubacar Keita and Prime Minister Moussa Mara, along with other government officials. It said she also toured the country's Centre for Vaccine Development, where an experimental Ebola vaccine is in phase 1 clinical trials.
Chan said in the statement that Mali has responded quickly with contact tracing. "It is vital right now that WHO and all other United Nations organizations working in Mali support the government in maintaining this strong response until we are certain this outbreak is contained."
The WHO statement cited Mali's newly confirmed Ebola case and its identification of two suspected cases. It said health officials are tracking several hundred contacts of the recent Ebola cases.
An update today from the UN Mission for Ebola Emergency Response (UNMEER) said Mali's president invited Chan to help organize the country's response to Ebola and that the WHO delegation and Mali's government will conduct a joint analysis of the situation and assess the country's needs and how the international community can respond to them.
The UNMEER report also noted that the UN Development Program launched a project to improve sanitation and health around the Guinea border town of Kouremale, where thousands of people travel from Guinea to Mali each day.
MSF assessment cites several concerns
In its latest outbreak assessment, Doctors without Borders (MSF) said on Nov 21 that the virus in Sierra Leone is outpacing response efforts, despite a wealth of technical expertise and material support. Though governments including the United Kingdom and China and other groups have sent teams to build new treatment centers and some are slated to open soon, they won't be at full capacity until well into the new year.
MSF said bed capacity is still a critical issue. "Moreover, MSF is concerned that transit facilities, holding facilities, functioning healthcare facilities, and ambulances can all act as points of transmission."
In Guinea, MSF said, patient numbers are still high, and the number of new cases continues to rise. It said the capital, Conakry, saw a small increase in cases over the past week and that the MSF transit facility in Macenta over the past 2 weeks had to halt admission twice, because the clinic was full. The MSF case management center in Gueckedou is full and the situation in border districts is worrisome, especially the eastern ones, the group said. It added that patients often come from far away and are often very sick by the time they arrive at the facility.
Liberia's case numbers are dropping in Monrovia and some other parts of the country, which MSF said is a positive development, while warning that the epidemic is far from over. For example, it said it continues to admit new patients—42 of them last week. The group added that it is still worried about poor ambulance and referral systems, community surveillance, safe body management, and contact tracing.
The Cess River area of rural Liberia is reporting some Ebola cases, and a team has arrived there to launch contact tracing, conduct safe burials, distribute protection kids, and prepare a transit center, the report said.
Other developments
- Italy's health ministry said an Italian doctor contracted Ebola while working in Sierra Leone and will be airlifted to Rome for treatment, the AP reported today. The doctor works for a nongovernmental organization called Emergency and is Italy's first Ebola case-patient. He will be treated at Lazzaro Spallanzani National Institute for Infectious Diseases.
- A UN worker who was flown to France after contracting Ebola in Sierra Leone has recovered and was released yesterday from a Paris hospital, Reuters reported. Reports haven't listed the patient's name or nationality.
- The Ebola outbreak has crippled the delivery of treatment and care to people living with HIV in Liberia, according to today's UNMEER report. An estimated 30,000 people have HIV in Liberia, and before the Ebola epidemic, more than 70% of them had access to treatment at 144 HIV/AIDS care centers across the country. However, the shortage of health workers and fear of Ebola transmission have shuttered 60% of the facilities. UNMEER said Liberia's AIDS control program workers are going door-to-door in Montserrado County to deliver antiretroviral drugs or to refer people to mobile treatment centers to get their prescriptions refilled.
See also:
Nov 24 Merck press release
Nov 24 NewLink press release
Nov 24 Canadian Press story
Nov 22 AP story
Nov 24 WHO report
Nov 24 UNMEER report
Nov 21 MSF update
Nov 24 AP story
Nov 23 Reuters story