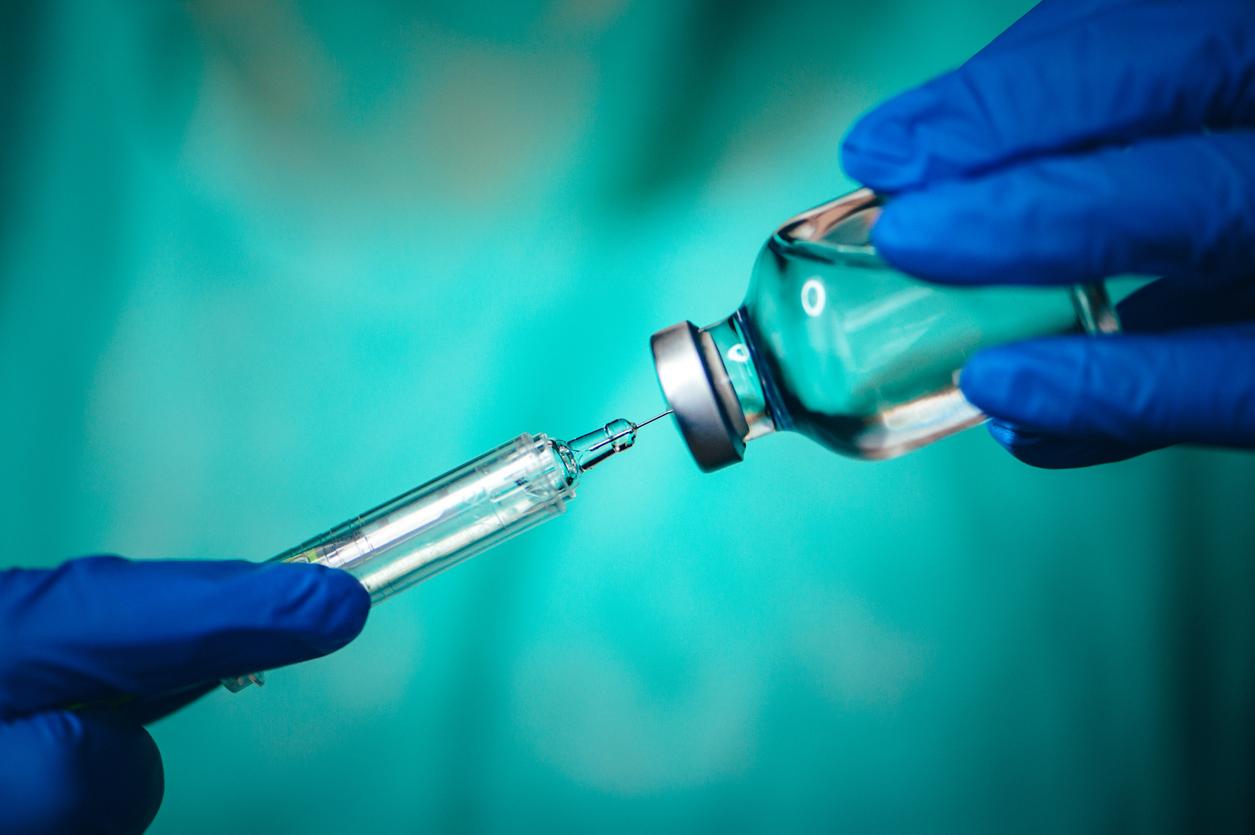
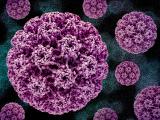
A new study from researchers at the Kenya Medical Research Institute (KEMRI) and Massachusetts General Hospital (MGH) showed a single dose of human papillomavirus virus (HPV) vaccine was highly efficacious in preventing HPV, the virus that causes cervical cancer, in girls and women ages 15 to 20.
The study results were announced at the 35th International Papillomavirus Conference in Washington, DC, this week.
Researchers described the findings from their randomized, multicenter, double-blind, controlled trial, which included 2,275 participants in Kenya between the ages of 15 and 20. The women were randomly assigned to receive either a single dose of the bivalent (two-strain) or nonavalent (nine-strain) HPV vaccine or the control vaccine, which contained two doses.
Participants were tested for HPV over a 36-month trial period, and both bivalent and nonavalent HPV vaccines were highly efficacious, with a vaccine efficacy of 98%, and 96%, respectively after only one dose. The findings build on researchers' previous work, which showed the vaccines were efficacious over an 18-month interval.
This study provides important evidence that a single dose of HPV vaccine can be highly effective in preventing persistent infections, and ultimately, cervical cancer
"This study provides important evidence that a single dose of HPV vaccine can be highly effective in preventing persistent infections, and ultimately, cervical cancer," said lead investigator Ruanne Barnabas, MD, of Massachusetts General Hospital in a statement on the study.
90% of cervical cancer deaths occur in poor countries
Of the estimated 342,000 cervical cancer deaths in 2020, more than 90% occurred in low- and
middle-income countries, the authors said. A single-dose vaccination schedule would mean more people could receive adequate protection from a simplified vaccine schedule.
In December 2022, the World Health Organization (WHO) updated guidance recommending one-dose coverage. Since then, eight countries have updated their vaccine schedule to reflect a single dose.
Countries that have yet to switch over have voiced concerns that a single dose is not durable enough.
"From our initial 18-month data, there have been concerns raised regarding the durability of the single dose HPV vaccine. This 36-month data helps to allay this anxiety," said Maricianah Onono, MD, a director of the study.