Mar 17, 2011
Expert calls for destroying smallpox stocks, limiting research on new vaccines
A US bioterrorism expert argues that, because priority should be placed on ensuring adequate supplies of existing smallpox vaccine and completing work on two key antivirals, maintaining reserves of smallpox viruses in the United States and Russia is no longer warranted. Writing in Biosecurity and Bioterrorism, D.A. Henderson, MD, MPH, of the UPMC Center for Biosecurity in Baltimore, contends that investing in new smallpox vaccines is prohibitive. He estimates the cost of developing a new third-generation vaccine at $250 million to $1 billion, plus $500 million to $750 million for a new manufacturing facility. In addition, developing a stockpile to vaccinate just 5% of the global population with current second-generation vaccines would cost $1 billion, while a global stockpile of nonreplicating (and therefore safer) third-generation vaccines for the same number of people would cost $8 billion. Henderson also points out that the most advanced third-generation smallpox vaccine, Imunogen, requires two doses 1 month apart and does not confer protection until 6 weeks after the first injection, which limits its utility for a bioterror attack. He calls for investments in strengthening surveillance, containment, and lab capabilities.
Mar 16 Biosecur Bioterror commentary abstract
Polio elimination efforts still ongoing in 3 African nations
Scientists from the US Centers for Disease Control and Prevention (CDC) today reported that efforts to stem the reintroduction of wild poliovirus (WPV) into four African nations are succeeding in only one of them, Sudan. Writing in Morbidity and Mortality Weekly Report (MMWR), the CDC experts say that, since 2002, outbreaks have occurred in 39 previously polio-free countries, but most of these were stopped in less than 6 months. However, circulation in Angola, Chad, the Democratic Republic of the Congo, and Sudan persisted longer than a year. The Global Polio Eradication Initiative aimed to eliminate WPV in those countries by the end of 2010, a goal that has been met only in Sudan, where no cases have been detected since June 2009. Efforts to eliminate the disease in the other three countries continue.
Mar 18 MMWR report
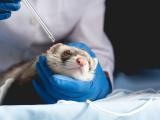
H5N1 spreads from digestive tract to multiple organs in study
In a study to determine how the deadly H5N1 avian influenza virus spreads to the respiratory tract, researchers found that when the virus was directly inoculated into the stomach of ferrets and hamsters, within a day it had spread to multiple organs, including the lung, liver, and brain. Histopathologic analysis showed lymph folliculitis in the digestive tract and mesenteric lymph nodes, as well as pneumonia. The authors write, "We conclude that, in mammals, ingested H5N1 influenza viruses can disseminate to non-digestive organs, possibly through the lymphatic system of the gastrointestinal tract."
Mar 16 J Virol abstract
Modified West Nile virus vaccine shows promise
A trial of a modified DNA-based vaccine against West Nile virus (WNV) in adults showed that it produced a stronger immune response than the first version of the vaccine and was equally effective in older people, the group at greatest risk for WNV infections and complications. The study, funded by the National Institute for Allergy and Infectious Diseases, was published early online by the Journal of Infectious Diseases. The DNA vaccine tested uses a configuration like that of a DNA vaccine that has already been cleared for use in horses. An earlier human trial of a similar vaccine showed that it was safe and immunogenic, and the current trial was designed to test a newer generation of the vaccine that includes a promoter to enhance immunogenicity. The trial included 30 healthy adults aged 18 to 65. A three-dose regimen was well tolerated and was more immunogenic than the first generation of the DNA vaccine. Antibody responses in the oldest half of the subjects were similar to those in the younger group. The researchers concluded that the promoter was effective in boosting the response to the vaccine and that it was safe and effective in older people. Currently, there are no WNV vaccines licensed for humans.
Mar 11 J Infect Dis abstract