Mar 8, 2013
Quadrivalent flu shot trial shows promising benefits for kids
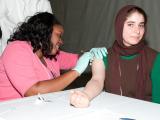
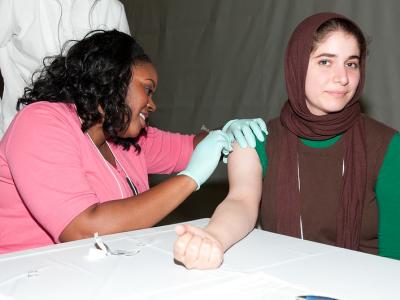
A study in children that compared an inactivated quadrivalent influenza vaccine (QIV) against two trivalent inactivated vaccines containing different flu B strains found the QIV more immunogenic against the unmatched B strain. The randomized, double-blind trial involving GlaxoSmithKline's QIV candidate appeared in the Journal of Infectious Diseases. The results are based on evaluation of 2,738 children 3 to 17 years old in the Czech Republic, France, Germany, the Philippines, and the United States from Oct 2010 to June 2011. Researchers also assessed QIV in a separate group of children ages 6 through 35 months old. After randomizing children to QIV or one of the TIV groups, researchers obtained blood samples 28 days after vaccination and conduced hemagglutination-inhibition tests to assess immunogenicity. They found that QIV was as immunogenic as TIV for the shared strains and superior to TIV against the unmatched strains. The arm of the study in babies and toddlers suggested that QIV was immunogenic against all four strains. Reactions and safety markers were similar in QIV and TIV groups. The researchers noted that the main limitation of the study is that immunogenicity doesn't predict the magnitude of protection against influenza B and said effectiveness trials are needed after the vaccine is used in campaigns. They concluded that there is a clear need for a quadrivalent vaccine in kids, because they bear a bigger burden if influenza B infections and that QIV may provide better protection against the strain than current vaccines.
Mar 7 J Infect Dis abstract
Study: Surgical masks may cut flu virus shedding by two thirds
Fine aerosolized particles carry nine times more influenza viruses than do coarser airborne particles, and having flu patients wear surgical masks may cut viral shedding to one-third that of unmasked patients, according to a study yesterday in PLoS Pathogens. US and Hong Kong researchers collected exhaled particles from 37 volunteers who were confirmed to have influenza by polymerase chain reaction. Samples were collected with the patients wearing surgical masks and without. The investigators found that fine airborne particles of 5 micrometers or less in diameter harbored 8.8-fold more viral RNA copies than did larger airborne particles. They also found that the masks reduced viral load 2.8-fold in the fine particles and 25-fold in coarser particles. Overall, masks reduced viral shedding 3.4-fold, or about 71%. The authors conclude, "These results suggest an important role for aerosols in transmission of influenza virus and that surgical facemasks worn by infected persons are potentially an effective means of limiting the spread of influenza."
Mar 5 PLoS Pathog study
USDA extends deadline for assessing ground-poultry safety plans
In response to an industry request, the US Department of Agriculture (USDA) announced yesterday that poultry processors have another 45 days—until Apr 20—to reassess their safety plans for raw ground chicken and turkey products. The USDA's Food Safety and Inspection Service (FSIS) also said processors have until the same date to comment on the reassessment order and related proposed actions, according to Federal Register notice. The FSIS announced on Dec 6, 2012, that processors had until Mar 6 to reassess their HACCP (hazard analysis and critical control point) plans for raw chicken and turkey products that are ground or "otherwise comminuted"—a step that was prompted by several recent Salmonella outbreaks linked to such products. In the December notice the FSIS also announced the expansion of its Salmonella testing program to all forms of raw comminuted poultry products not destined for further processing into ready-to-eat products. In January a coalition of trade groups asked the agency for more time to comment on the plans, according to the Federal Register notice. While granting the request, the FSIS said it plans to start taking samples of ground poultry for Salmonella testing on or around Apr 20.
Mar 7 FSIS Federal Register notice
Related Dec 6, 2012, CIDRAP News story