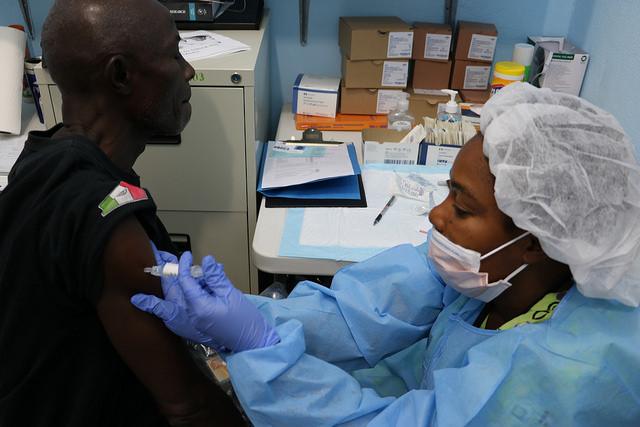
Two more Ebola infections have been confirmed in Democratic Republic of Congo (DRC) Ebola outbreak, both from Beni, which has been the outbreak's main hot spot over the past several weeks.
Also today the World Health Organization (WHO) said four of the most recent cases involved healthcare workers, and in research developments, scientists yesterday reported durable immune response for three Ebola vaccines, including the one currently deployed in the DRC.
Beni accounts for nearly half of all outbreak cases
The pair of newly confirmed cases reported from Beni lift the overall outbreak total to 276, which includes 241 confirmed and 35 probable infections, according to the health ministry's daily update.
Of the case total, nearly half (134) are from Beni, where responders have grappled not only with security problems due to conflict between rebel groups and armed forces, but also pockets of community resistance.
One new death was reported, which involved a patient from Beni. That development boosts the outbreak fatality total to 175 deaths.
The number of suspected cases has jumped to 65, up from 32 the previous day.
Responders continue to make headway with vaccination, and 24,510 people have now been immunized with VSV-EBOV, roughly half of them from Beni.
More health worker infections
In its weekly situation report today, the WHO said it has received 36 more illness reports since its last update on Oct 23. It said there are four new cases among health workers, one doctor and three nurses. All worked in health centers around Beni, raising the number of infected medical providers to 25, including 3 deaths.
The percentage of contacts successfully followed over the past week was 92% for all health zones and 91% for Beni. So far, 109 patients have received experimental treatments.
Though health officials are still concerned about security for burial teams, activities have resumed in Butembo for the first time since an Oct 2 security incident.
The number of confirmed cases in Beni and in and around Butembo continues to increase, and lingering security and community resistance challenges highlight the need to press ahead with community engagement activities, the WHO said.
The spurt in health worker infections is concerning and shows that infection prevention and control measures need to be stepped up in the affected areas, it added.
Lasting antibody response from 3 vaccines
On the research front, at the American Society of Tropical Medicine and Hygiene (ASTMH) annual meeting under way in New Orleans yesterday, researchers from the United Kingdom said the immune response from three Ebola vaccines, including VSV-EBOV, lasts at least 2.5 years.
The other two were prime-boost regimens that used one of two nonreplicating adenoviruses (ChAd3 and AdHu26) and the multivalent MVA BN-Filo, according to the meeting abstract. ChAd3 was developed by the National Institutes of Health and GlaxoSmithKline and AdHu26 was developed by Janssen and is part of Johnson and Johnson's candidate vaccine. MVA BN-Filo, developed by Bavarian Nordic, contains glycoproteins from Ebola Zaire and Sudan strains and Marburg virus.
The researchers based their findings on an analysis of blood samples from healthy volunteers who had taken part in earlier research studies involving three vaccines. So far, this is the longest researchers have been able to track Ebola vaccine response.
For their the prime-boost vaccines, the team looked at the durability of both humoral and cellular immunity. They found that 91% of 43 people who received the AdHu26 vaccine had positive glycoprotein-specific IgG titers 2.5 years after immunization, as did 54% of 13 who received the ChAd3 vaccine, according to the meeting abstract.
In a press release from ASTMH, Katie Ewer, PhD, with the Jenner Institute at the University of Oxford, said all three vaccines "were still producing a strong antibody response to the disease two and a half years after immunization, which is really good news."
She said another important question is if persistence can be enhanced by giving a late booster dose 3 to 4 years after initial immunization and that the group will study this in the United Kingdom and Senegal in the year ahead.
See also:
Oct 30 DRC update
Oct 30 WHO situation report
Oct 29 ASTMH press release
Oct 29 ASTMH study abstract