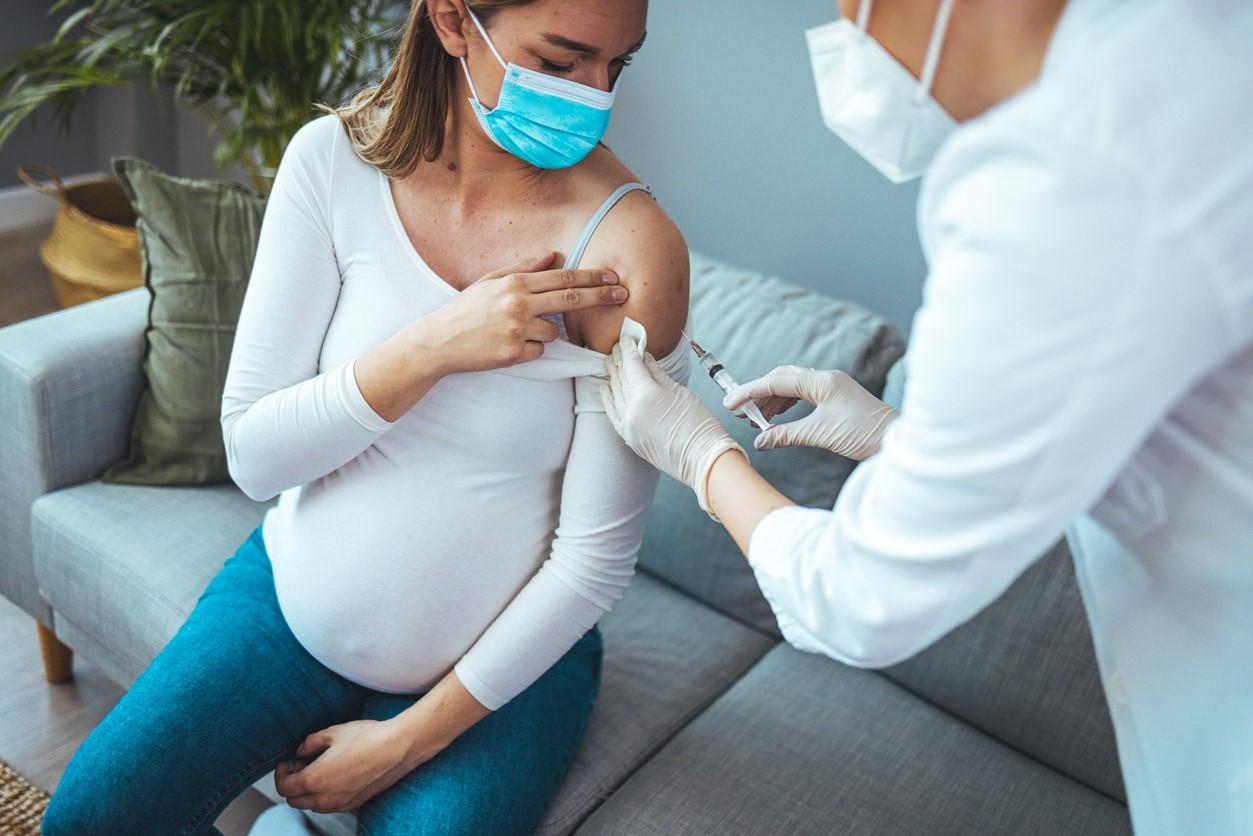
The Food and Drug Administration's (FDA's) vaccine advisory group yesterday recommended Pfizer's respiratory syncytial virus (RSV) vaccine as a tool to protect newborns, though the group voiced some uncertainty about safety.
Twists and turns on the RSV vaccine path
Infants and older people are at the highest risk for severe RSV, and two RSV vaccines—one from Pfizer and the other from GlaxoSmithKline—have gone through clinical trials and are making their way through the FDA's regulatory process.
Earlier this month, the FDA approved GSK's vaccine for use in people ages 60 and older, bringing the nation a way to protect the older risk group. The strategy for protecting newborns and younger infants revolves around vaccinating pregnant women. The Centers for Disease Control and Prevention (CDC) estimates that 58,000 to 80,000 children under age 5 are hospitalized with RSV each year and that 1 to 2 of every 100 children younger than 6 months old with RSV may need to be hospitalized.
In late February, the advisory group narrowly recommended Pfizer's vaccine for the older age-group, over concerns about Guillain-Barre syndrome and a desire to see more data. The vaccine still awaits full FDA approval. And in February 2022, GSK stopped its RSV trials in pregnant women to better analyze safety data.
Qualms about preterm births
After a full day of discussion, hearing from the company, FDA experts, and members of the public, the FDA's Vaccines and Related Biological Products Advisory Committee (VRBPAC) voted separately on efficacy and safety questions.
The positive recommendation was unanimous for efficacy. A large study from Pfizer suggested that the efficacy was 82% for preventing severe RSV in babies from birth to 3 months old when administered to the mother during the second or third pregnancy trimester. At age 6 months, efficacy was still high, at 69% against severe RSV illness.
Regarding safety, the vote to recommend the vaccine passed on a narrower 10-to-4 vote. Studies revealed no concerns, though there was a slight increase in preterm births in the RSV vaccine group that wasn't statistically significant. At 5.7%, the rate of preterm births in vaccinated women was slightly higher than the 4.7% in the unvaccinated group. Also, the preterm births that occurred in the pregnant women occurred late in the gestational period.
Though the GSK vaccine wasn't part of the consideration for the Pfizer vaccine, the fact that the company stopped pursuing it for use in pregnant women loomed large over yesterday's discussions. Several VRBPAC members said more data are needed to clarify preterm birth questions, and the group agreed that postmarketing data will provide crucial information on the issue.
Paul Offit, MD, professor of pediatrics at the Children's Hospital of Philadelphia, said, "A lot's at stake. We're risking one group to protect another, and there's not enough data to be reassured." Other members raised concerns about unanswered questions regarding vaccine coadministration in pregnant women.
A lot's at stake. We're risking one group to protect another.
The group, however, was impressed by the vaccine's potential impact, especially against severe disease. Jay Portnoy, MD, professor of pediatrics at Children's Mercy Hospital in Kansas City, Missouri, said if the vaccine lives up to its promise, "parents can breathe easier."