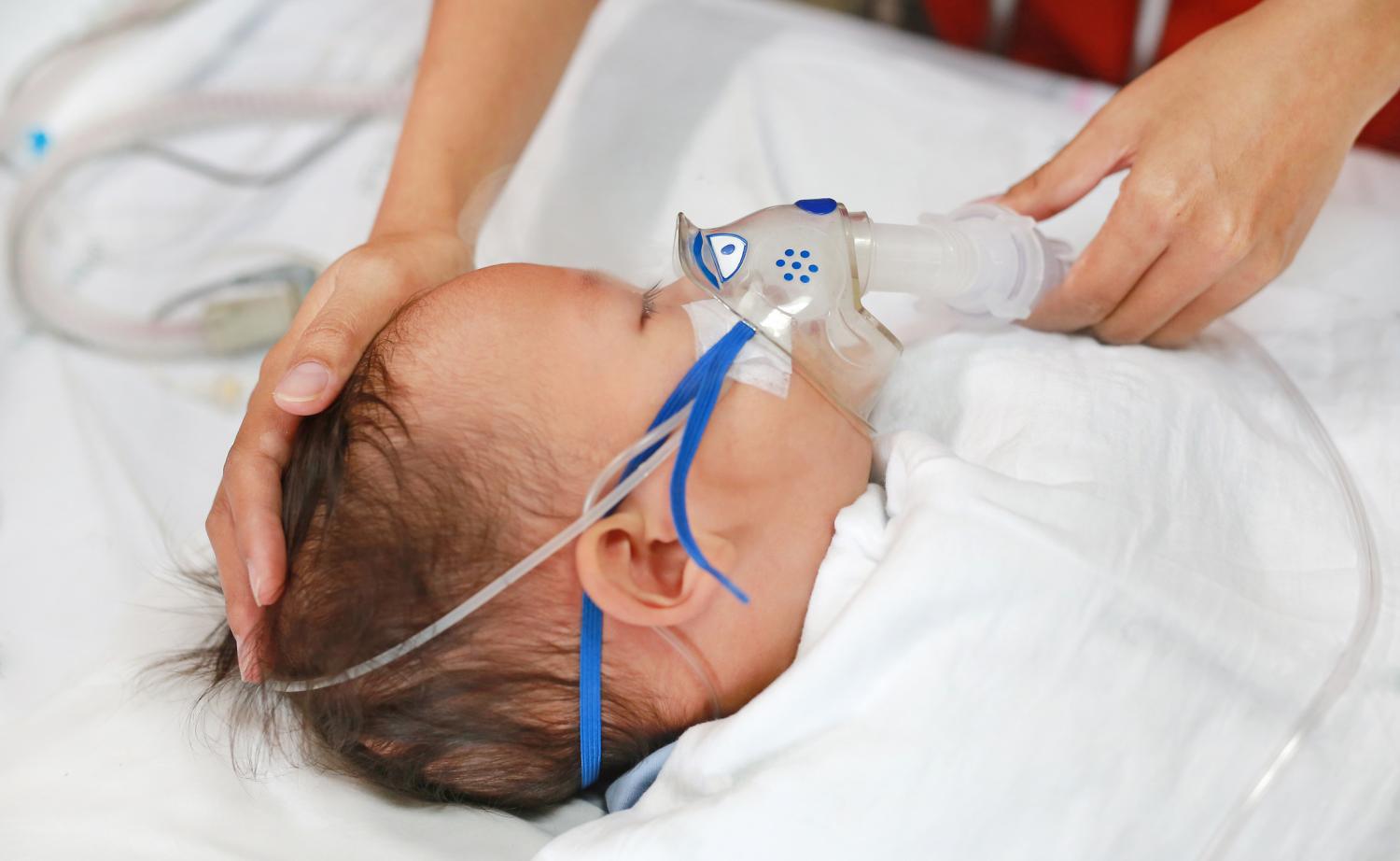
Two studies highlight new respiratory syncytial virus (RSV) findings, with one estimating that the monoclonal antibody nirsevimab was 80% effective against hospitalization in French pediatric intensive care units (PICUs) and the other showing that more hospitalizations occurred during the atypical 2021 and 2022 RSV seasons despite similar disease severity to previous seasons.
RSV is the No. 1 cause of the hospitalization of US infants, leading to an estimated annual 58,000 to 80,000 hospitalizations and 100 to 300 deaths in children younger than 5 years old. Risk factors for severe disease include younger age and prematurity.
Estimated 80% effectiveness against severe illness
For the first study, published yesterday in Influenza and Other Respiratory Diseases, public health researchers in France led an analysis of PICU network data to estimate the effectiveness of nirsevimab against severe RSV bronchiolitis from September 2023 to January 2024. The authors called it the first study to provide real-world estimates of nirsevimab effectiveness against severe RSV in PICU patients.
The country mounted a national immunization campaign in September 2023. The case-control study, which used a test-negative design, included 288 RSV-infected infants and 50 control patients in 20 PICUs; 91% were aged 0 to 3 months, and 55% were boys.
During the study period, 58 (20%) infants received nirsevimab at least 8 days before PICU admission (average delay, 35 days). These infants were more often premature than untreated infants (29% vs 10%). Estimated nirsevimab effectiveness was 75.9% in the main analysis and 80.6% and 80.4% in two sensitivity analyses, confirming results of clinical studies.
"Nirsevimab seems to be able to occupy a significant position in the therapeutic arsenal for preventing RSV infections, although it must be compared to other preventive treatments in development," the researchers concluded. "The RSV vaccine for pregnant women is actually under evaluation by health authorities and not yet available in France."
Higher hospitalization rates in kids 2 to 5 years in 2022
Today in Pediatrics, a study led by US Centers for Disease Control and Prevention (CDC) researchers reports on the RSV resurgence in 2021 and 2022 after an early-pandemic lull. "The coronavirus disease 2019 pandemic disrupted respiratory syncytial virus (RSV) seasonality resulting in early, atypical RSV seasons in 2021 and 2022, with an intense 2022 peak overwhelming many pediatric healthcare facilities," the study authors noted.
The researchers conducted RSV surveillance for acute respiratory illness among children younger than 5 years old at seven pediatric hospitals from 2016 to 2022. The team interviewed parents, reviewed medical records, and tested respiratory specimens for RSV and other respiratory viruses to determine the hospitalization rates and characteristics of infected children.
Nirsevimab seems to be able to occupy a significant position in the therapeutic arsenal for preventing RSV infections, although it must be compared to other preventive treatments in development.
The median age of children hospitalized for RSV was higher in 2022 (9.6 vs 6.0 months), and RSV hospitalization rates were higher in 2021 and 2022 than the prepandemic average across all age-groups.
RSV hospitalization rates were similar among children younger than 2 years in 2021 and 2022, but those aged 24 to 59 months had significantly higher rates of hospitalization in 2022 (rate ratio, 1.68).
"Lack of prior RSV exposure in children aged 12 to 23 months may have led to some increases in hospitalization rates in 2021 and 2022; however, the magnitude of the increase observed in older children in our analysis is substantially less than would have been expected if these same children had been exposed in infancy," the researchers wrote.
More children hospitalized for RSV needed supplemental oxygen, and more respiratory virus co-infections were detected in 2022 than before the pandemic, but the proportion of patients who had low oxygen levels, required mechanical ventilation, were admitted to intensive care remained the same.
"RSV prevention products have demonstrated efficacy against RSV disease, not infection," the authors said. "Our findings are reassuring and suggest that even if these products did prevent—and effectively postpone primary RSV infection—we would not see substantial increases in hospitalization rates in children 12 to 23 months of age after the introduction of maternal vaccination or widespread immunoprophylaxis [prevention] with long-acting monoclonal antibody products."