Across the country 7-day averages of new COVID-19 cases and hospitalizations are dropping in 43 and 49 states, respectively. According to CNBC, only Nebraska, New Hampshire, North Dakota, Vermont, and Wyoming reported a rise in new cases.
The US reported 69,230 new COVID-19 cases yesterday, and 2,542 deaths, according to the Johns Hopkins COVID-19 tracker. In total, the US remains the hardest hit country in the global pandemic, with 27,950,547 cases and 495,015 deaths.
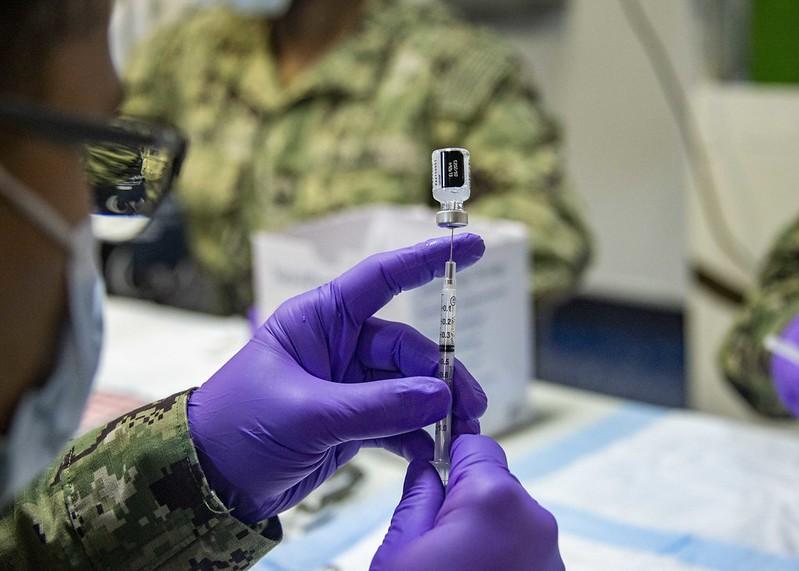
The news of case declines comes as Pfizer and BioNTech, the makers of the first mRNA vaccine approved for use in the United States, began a global phase 2/3 trial targeting pregnant women, and submitted new temperature data to the Food and Drug Administration (FDA) suggesting they could allow their COVID-19 vaccine to be stored in pharmacy freezers rather than ultra-cold storage facilities.
The drug maker said tests on the vaccine show it remained stable when stored at -25°C to -15°C (-13°F to 5°F), temperatures commonly found in pharmaceutical freezers and refrigerators. Currently, under the FDA's emergency use authorization of the Pfizer-BioNTech vaccine, the vials must be stored at -80ºC and -60ºC (-112ºF to ‑76ºF).
"Our top priority was to quickly develop a safe and effective vaccine and make it available to the most vulnerable people in the world in order to save lives. At the same time, we have continuously collected data that could enable storage at around -20°C. The data submitted may facilitate the handling of our vaccine in pharmacies and provide vaccination centers an even greater flexibility," said Ugur Sahin, CEO and co-founder of BioNTech. "We will continue to leverage our expertise to develop potential new formulations that could make our vaccine even easier to transport and use."
Yesterday, Pfizer announced it would begin testing its two-dose vaccine in 4,000 healthy pregnant women over the age of 18 vaccinated during 24 to 34 weeks of gestation. Women will be followed throughout pregnancies, and infants through the age of 6 months, Pfizer said.
Today President Joe Biden visited the Pfizer vaccine plant in Kalamazoo, Michigan. Biden met with workers who produce the vaccine and toured the facility.
Fauci calls Israeli study ‘interesting’
Today during a White House press briefing on COVID-19, chief medical adviser to President Biden, Anthony Fauci, MD, said he thought a new Israeli study showing that one dose of the Pfizer-BioNTech vaccine was 85% effective 15 to 28 days after inoculation was "interesting."
"These numbers do look interesting, but we don’t know how durable a single dose is," said Fauci. "We will stick with the scientifically documented efficacy of vaccine prime followed by a boost."
Fauci also commented that he believed both Pfizer and Moderna would have enough safety and efficacy data on their vaccines for use in teenagers sometime this fall, but he said data on elementary school students was unlikely to come before the first quarter of 2022.
Six million doses delayed due to weather
Andy Slavitt, the White House Senior Advisor for COVID response, today said that 6 million doses of vaccine have been delayed this week because of inclement weather across much of the United States, including Texas, which is experiencing severe power outages after a winter storm.
"We're asking vaccine administration sites to extend their hours even further and offer additional appointments and to try to reschedule the vaccinations over the coming days and weeks as significantly more supply arrives," Slavitt said during today's press briefing.
According to the Dallas Morning News, the Centers for Disease Control and Prevention (CDC) and shipping companies postponed last Friday's shipments, which included 407,000 first doses and 333,000 second doses to Texas, in anticipation of the bad weather.
CDC: First month of vaccine safety data shows few adverse events
Finally, today in the CDC's Morbidity and Mortality Weekly Report, authors describe the first month of safety data of the Pfizer-BioNTech and Moderna vaccines in the United States collected through the Vaccine Adverse Event Reporting System (VAERS).
Between Dec 13 and Jan 14, 13,794,904 vaccine doses were administered, and VAERS received and processed 6,994 reports of adverse events after vaccination, including 6,354 (90.8%) that were classified as nonserious and 640 (9.2%) as serious, the report said. The most common adverse symptoms were headache (22.4%), fatigue (16.5%), and dizziness (16.5%).
One hundred and thirteen deaths were reported, but vaccination was not ruled as cause of death for anyone.
"These data provide reassurance and helpful information regarding what health care providers and vaccine recipients might expect after vaccination," the report said. "Counseling vaccine recipients to expect transient local and systemic reactions might ease concerns and encourage completion of the 2-dose vaccination series."