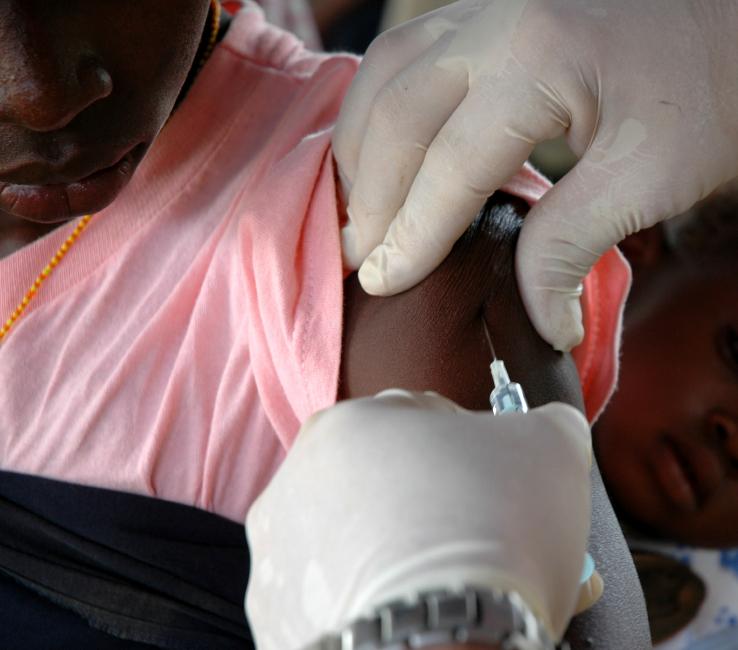
The next steps in testing Ebola vaccines as well as a picture of how they might be used in West Africa's outbreak region became clearer today as officials from the World Health Organization (WHO) shared the outcome of yesterday's high-level meeting in Geneva.
Large phase 3 trials in West Africa's outbreak setting will be next up to see if the vaccines work, with the studies designed to collect more safety data, a task usually covered in phase 2 trials, experts involved in the meeting said today at a WHO media telebriefing. However, as Ebola infections drop off in the outbreak region, scientists face a closing window of opportunity to see how the vaccines perform in a real-life setting.
Experts also signaled a possible role for two Ebola vaccine formulations: a single-dose vaccine providing shorter-term immunity that could be used to tamp down Ebola flare-ups and a prime-boost vaccine that may provide more durable immunity and could be used for other immunization strategies.
Vaccine landscape evolves
Marie-Paule Kieny, PhD, the WHO's assistant director-general for health systems and innovation, said the group's review of phase 1 clinical trials of the two vaccines furthest along in testing found acceptable safety profiles, but information on what dose stimulates an ideal immune response won't be known for 2 to 4 more weeks.
While 2014 will be remembered as the year Ebola challenged humanity, 2015 will be remembered as the year humanity used the best scientific minds to fight back against the disease, she said, and the fast-tracking of the vaccines has been unprecedented. "I can assure you there has been no holiday for the many people working on that."
Helen Rees, MBBS, a professor at the University of Witwatersrand who led the meeting, told reporters that progress made at the meeting was very encouraging and that each of the affected countries had strong delegations that were deeply engaged.
Yesterday's meeting was the second to work out details about Ebola vaccine clinical trials and how to finance eventual vaccine deployment to curb spread of the virus in Guinea, Liberia, and Sierra Leone. Since the first meeting on Oct 23, a third Ebola vaccine has entered phase 1 trials: a prime-boost combination from Johnson & Johnson, based on AdVac technology from Crucell Holland BV and MVA-BN technology from Bavarian Nordic.
Clinical trials have been under way for the past few months for the two other vaccines: a US-GSK vaccine known as ChAd3 that uses a chimpanzee adenovirus with a Zaire Ebola virus gene spliced into it, and VSV-EBOV, developed by Canadian researchers and licensed by NewLink Genetics and Merck, which uses a vesicular stomatitis virus (VSV) to carry an Ebola gene.
Developers of the Johnson & Johnson vaccine, for which phase 1 trials just launched, have overcome some delays and successfully gained some ground, Kieny said, adding that additional Ebola vaccines, including ones from groups in Russia and China, are at different stages of development. "Last year at this time the cupboard was dry, but now the cupboard is filling quickly," she said.
The next phase of testing will take place in all three West African countries, using three completely different trial designs, which Kieny said will give researchers several good points for evaluating the vaccines.
Phase 3 study details
A randomized trial in Liberia will involve three arms (single doses of ChAd3, VSV-EBOV, and placebo), with 9,000 volunteers in each. In Sierra Leone, investigators will conduct a trial of a still-undetermined vaccine using a step-wedge design, with a goal of enrolling about 6,000 people. Meanwhile, teams in Guinea will vaccinate frontline health workers at trial sites where they will use a ring-vaccination design involving about 4,500 people, similar to an intervention used to vaccinate against smallpox.
Kieny said researchers hope to begin the Liberian trial at the end of January, with trials in Sierra Leone and Guinea projected to begin in February. She said efforts—such as sensitizing the population—are already under way to prepare for the vaccine trial. Study sites are getting special freezers to store the vaccine, but she added that developers are working on formulations that potentially allow the doses to be stored at refrigerator temperatures and that might be available in the next few months.
Experts at the meeting discussed concerns about how a continued drop-off in cases could affect the trials, Kieny said. She noted that investigators could include larger populations if they need to, though doing so might lead to some delays.
Weighing vaccine choices, outbreak needs
Looking ahead to production capacity for the three vaccines that are furthest along in trials, Kieny said robust quantities in the tens of millions are expected for VSV-EBOV as well as for Johnson & Johnson's prime-boost vaccine, which can be stored at refrigerator temperature. However, the quantity for ChAd3 will depend on the dosage level, with a few million doses available by the middle of the year.
Rees said a separate WHO working group is exploring different strategies for deploying vaccines once they are available. Immunization plans will account for different epidemiologic patterns in each of the countries.
When asked if all the vaccine studies were taking up too much research space, Rees said there is capacity to do more than one phase 3 study and that having several vaccine candidates in the pipeline is desirable. "We can't assume that anything is going to work," she said, adding that once correlates of protection are known, scientists will look at how to improve the products.
She said the experts have addressed whether the vaccines will really make a difference in West Africa's outbreak. Different epidemiologic patterns in the region will require different control approaches, and even if vaccines have limited utility in the current outbreak, they will be ready for the future. "We anticipate that this won't be the end of Ebola."
Other developments
- In the wake of West Africa's Ebola outbreak and criticism of slow international response, 95 members of the global health academic community signed on to a letter published in The Lancet today urging countries to redouble their efforts to invest in the WHO. They said an analysis of how the group performed in the outbreak is warranted, but added that an independent review should also consider the impact of budget cuts to the WHO and the need for sustained funding. They said international forums are floating the idea of a new "first responder" United Nations agency but that the WHO already has a system in place and any new agency would face the same funding challenges.
- Urgent studies should be done to explore possible benefits of loperamide use, in combination with oral rehydration, for treating diarrhea in Ebola patients, three US-based researchers wrote yesterday in a commentary in the Journal of Infectious Diseases. In West African settings that lack the ability to provide electrolyte monitoring and replacement, antidiarrheal agents may be useful, but their use isn't often reported and there are no safety and efficacy data to guide their use. Clinicians may be reluctant to use the drug, because it hasn't shown a benefit for cholera-related diarrhea and can result in problems when used for some bacterial causes of diarrhea. However, they said studies have shown a benefit for other types of infectious diarrhea in adults and children older than age 2, with few safety problems. Use of loperamide seems like a rational approach that could also reduce environmental contamination, making it worthy of more study, the authors wrote.
- The number of Ebola infections in the three most affected countries has risen to 21,086, with the number of deaths increasing to 8,289, the WHO said today in a brief update. The totals reflect an increase of 114 illnesses and 30 fatalities since the WHO's update yesterday. Today's count includes cases reported in Liberia and Sierra Leone through Jan 6 and in Guinea through Jan 7. With 10,030 illnesses reported to far, Sierra Leone remains the hardest hit country. However, Liberia still has the most deaths, with 3,515.
See also:
WHO background on Jan 8 high-level meeting on Ebola vaccines
Jan 9 WHO press briefing audio file
Jan 10 Lancet letter
Jan 8 J Infect Dis abstract
Jan 9 WHO situation update