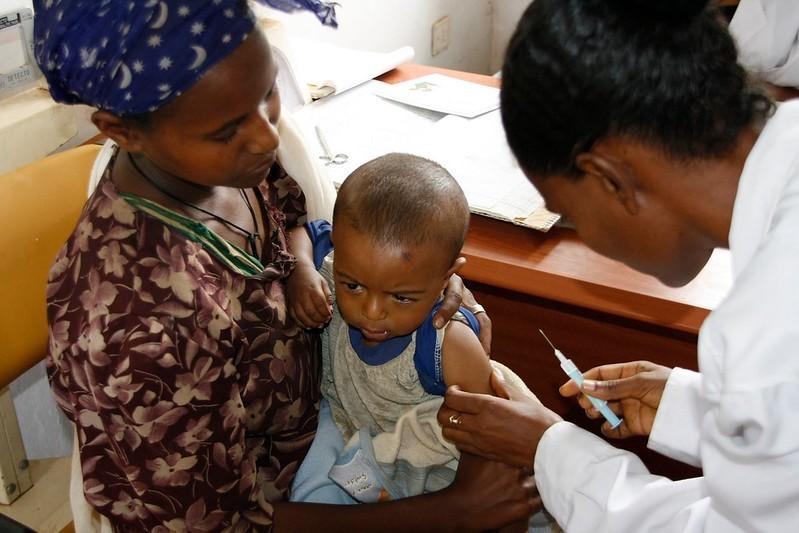
Global plans to cut the malaria burden in African children with the new RTS/S vaccine from GSK took a major step forward today with the World Health Organization (WHO) and UNICEF announcing that 18 million doses will be allotted to 12 countries, including 9 that haven't received it before.
Malaria is still one of Africa's deadliest diseases. In 2021 alone, the mosquito-borne parasite led to the deaths of about 500,000 children younger than 5 in Africa. GSK's RTS/S vaccine has been in the making for more than 30 years, launching in Malawi in 2019 as part of a pilot program that currently includes Ghana and Kenya.
At a WHO media briefing today, Director-General Tedros Adhanom Ghebreyesus, PhD, said with changing weather patterns amid the climate crisis, "mosquitoes that carry these diseases are increasing in density and spreading further afield." He added that 1.6 million doses of the vaccine have been given in the three pilot countries. "It has been shown to be safe and effective, resulting in a substantial reduction in severe malaria and a fall in child deaths."
The WHO prequalified the vaccine in July 2022. With limited supplies of the new vaccine, the agency also launched a framework for allocating supplies, which with input from countries, experts, and other health partners, took into account countries at highest risk and how interested nations in the region are incorporating RTS/S into their routine childhood immunizations.
High interest from African countries
In a statement today, the WHO and UNICEF said at least 28 African countries have expressed interest. And at today's briefing, Tedros said health officials are heartened by the region's high interest in receiving the vaccine.
Of 18 million available doses available for the next 3 years, global health officials allocated doses for the pilot programs under way in Malawi, Ghana, and Kenya. The nine countries that will receive their first shipments include Benin, Burkina Faso, Burundi, Cameroon, the Democratic Republic of the Congo, Liberia, Niger, Sierra Leone, and Uganda.
We will work tirelessly to increase supply until all children at risk have access.
Thabani Maphosa, MD, managing director of country program delivery at Gavi, the Vaccine Alliance, said in the WHO statement that while manufacturers ramp up supply, the goal is to ensure that the doses are used as efficiently as possible while applying lessons learned from the pilot programs.
Families and communities rightly want the vaccine for their children, said Kate O'Brien, MD, the WHO's director of immunization, vaccines, and biologicals. "The high demand for the vaccine and the strong reach of childhood immunisation will increase equity in access to malaria prevention and save many young lives. We will work tirelessly to increase supply until all children at risk have access."
Growing demand, next steps
The WHO estimates that annual demand for the vaccine will be at 40 million to 60 million by 2026 and will grow to 80 million to 100,000 million a year each year by 2030. It added that GSK is producing the vaccine and that India's Bharat Biotech will supply doses in the future.
Also, a second malaria vaccine is expected to enter the market. The WHO said it may soon quality the R21/Matrix-M vaccine, developed by Oxford University and made by the Serum Institute of India. In April, Ghana became the first country to approve the R21/Matrix-M vaccine.