Nov 8, 2011
Feds to conduct first national EAS test tomorrow
Federal agencies tomorrow will conduct the first nationwide emergency alert system (EAS) test tomorrow at 2:00 p.m. Eastern Standard Time, the Federal Emergency Management Agency (FEMA) said today. The test will air over all broadcast radio and television stations, cable television, satellite radio and television systems, and wireline video service systems for 30 seconds. It will not involve Internet services or cell phones. The test is designed to allow FEMA and the Federal Communications Commission to determine how well the system would function during a national emergency, such as a tsunami or a terrorist attack. In a recent letter to partners, FEMA administrator W. Craig Fugate said the first step in building a modernized EAS is to test existing technology, which has already identified gaps in the 50-year-old system not built to support closed captioning and various audio features. He warned that though the test will resemble routine local tests, the text on television screens may identically match the audio message on every television channel. A 2006 executive order requires the government to have an EAS for the President to use to provide critical information to the public during an emergency.
Nov 8 FEMA press release
FEMA letter
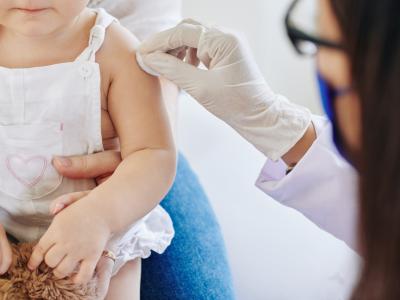
Vaccine-induced antibodies to 2009 H1N1 virus lasted a year in many children
In a Canadian study, close to half of children who received a single dose of an adjuvanted pandemic 2009 H1N1 influenza vaccine in 2009 still had what was considered a seroprotective level of antibodies to the virus a year later, according to a report published yesterday in Vaccine. In the fall of 2010, the researchers assessed the persistence of antibodies to the pH1N1 vaccine and the effects of the 2010-11 seasonal trivalent inactivated vaccine (TIV) in children between 15 months and 12 years old. At two pediatric clinics in Quebec City, they enrolled 128 children who had received the ASO3-aduvanted monovalent H1N1 vaccine a year earlier. Before and after the children received one or two doses of the 2010-11 seasonal vaccine, hemagglutination inhibition was used to check the children's antibody levels to the vaccine components: the 2009 H1N1 virus, a 2009 H3N2 strain, and a 2008 influenza B strain. Before the children received the 2010-11 vaccine, 46% of them had seroprotectoin (antibody titer of 40 or higher) against the 2009 virus, with only 37% and 19% showing protection against the H3N2 and B strains. After one dose of the seasonal vaccine, seroprotecton levels to H1N1, H3N2, and B reached 98%, 75%, and 57%. For children who received a second dose, the levels for H3N2 and B rose to 96% and 87%. The authors say their finding of substantial pH1N1 antibody persistence, along with high H1N1 immunization coverage in Quebec and the boosting effect of the 2010-11 vaccine, may help explain the lack of pH1N1 activity in Quebec in the 2010-11 season. However, they caution, "It should be recognized that serologic correlates of protection in children have not yet been established and some inherent variation in assay results based on laboratory methods is acknowledged."
Nov 7 Vaccine abstract
Thailand warns of flood-related leptospirosis
Widespread flooding in Thailand has prompted officials to warn of leptospirosis and other waterborne and foodborne diseases, according to Bangkok-based The Nation today. The Public Health Ministry warned of possible leptospirosis in flooded areas after it reported one confirmed and 20 suspected cases of the bacterial disease in Khon Kaen. The agency also warned against consuming ice from unknown sources. A recent survey reported contaminated food and water in four flooded provinces, including Bangkok, according to the story. The capital city is able to collect only 30% of its daily garbage because of the floodwaters.
Nov 8 Nation story
Georgia Tech to study microneedle patch for polio vaccine
Georgia researchers were awarded funding by the Bill & Melinda Gates Foundation (BMGF) to test the feasibility of microneedle patches to deliver polio vaccine cheaply. The Georgia Institute of Technology will receive funding through the BMGF's Grand Challenges Explorations, an initiative that promotes testing of unorthodox ideas to address persistent health problems, according to a Georgia Tech press release yesterday. Georgia Tech will collaborate on the project with researchers from the US Centers for Disease Control and Prevention. The current oral polio vaccine is very effective but uses a weakened virus and can cause the disease, according to the release, while injected vaccines require refrigeration and are expensive. The patch may offer a low-cost alternative using a killed virus if it is found effective. "Our goal for this study will be to provide the data to scientifically justify moving the microneedle patch for polio vaccination into a human trial," said lead researcher Mark Prausnitz of Georgia Tech.
Nov 7 Georgia Tech press release
FDA launches Web site on proper disposal of needles, other 'sharps'
The US Food and Drug Administration (FDA) today launched a Web site for patients and caregivers covering the safe disposal of hypodermic needles and other medical "sharps" at home, at work, and while traveling, the agency said in a news release today. Sharps are any medical devices that can puncture or cut the skin. The FDA said the new site "will help people understand the public health risks created by improperly disposing of used sharps and how users should safely dispose of them."
Nov 8 FDA news release
FDA "sharps" Web site