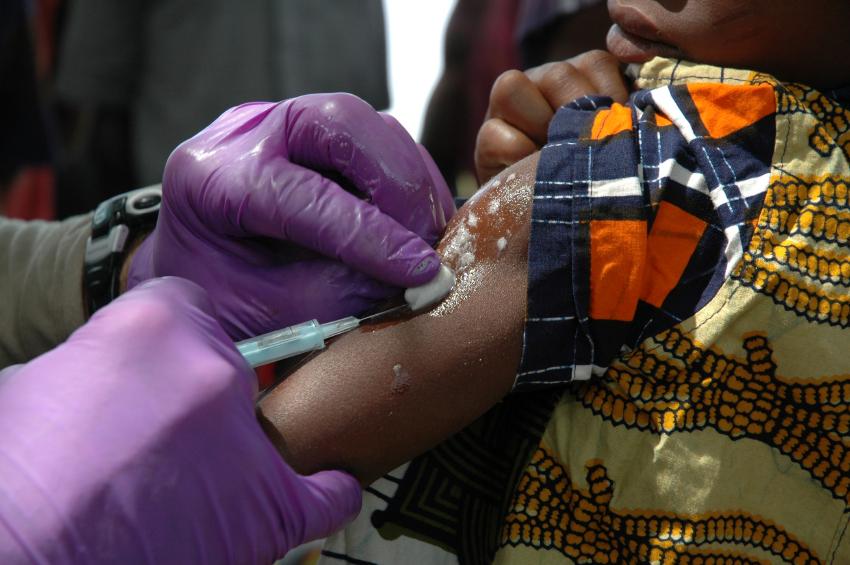
At a wide-ranging symposium on West Africa's Ebola epidemic today, much of the attention focused on the hope of an effective vaccine, as a US official announced that a large clinical trial of two candidate vaccines may be launched in Liberia in December.
Peter Jahrling, PhD, of the National Institute of Allergy and Infectious Diseases (NIAID), announced that plans call for a trial involving 30,000 participants, with 10,000 getting one of two candidate Ebola vaccines and 10,000 receiving a hepatitis vaccine as a control.
Calling the project a phase 2/3 trial, he said, "One would hope that once the results are analyzed it would facilitate licensure of at least one of those." He is chief scientist at the NIAID's Integrated Research Facility.
Also at the symposium, Michael T. Osterholm, PhD, MPH, of the University of Minnesota said his Center for Infectious Disease Research and Policy (CIDRAP) will collaborate with Jeremy Farrar, MBBS, DPhil, director of the UK-based Wellcome Trust, to prepare a report detailing the challenges that must be met to deliver an effective Ebola vaccine.
Calling a vaccine the only real hope for stopping the Ebola epidemic, he said the aim is to produce the document within 60 days. CIDRAP is the publisher of CIDRAP News.
The half-day symposium was sponsored by Johns Hopkins University's Bloomberg School of Public Health in Baltimore and was streamed over the Web.
Vaccines and treatments
In a brief presentation on candidate Ebola vaccines and drugs, Jahrling said one vaccine to be tested in Liberia is ChAd3, for chimpanzee adenovirus 3, developed by NIAID in collaboration with vaccine maker GSK. It uses the adenovirus as a carrier of pieces of Ebola virus genetic material.
The vaccine provided 100% protection in nonhuman primates and is now in phase 1 testing in the United States, he said. GSK, he added, predicts that 15,000 doses will be available by December.
The other vaccine to be tested is one based on a vesicular stomatitis virus (VSV) and was developed by the Public Health Agency of Canada 10 years ago, Jahrling said. It is now licensed to NewLink Genetics of Ames, Iowa. He said it was 100% protective in primates after 21 days, and it was used on the victim of a lab accident in Germany several years ago.
Jahrling noted that other Ebola vaccines are being developed by Bavarian Nordic, Crucell, and the US Army Research Institute of Infectious Diseases.
Discussing candidate drugs, Jahrling said ZMapp, a monoclonal antibody preparation made by Mapp Biopharmaceuticals, has now been given to seven Ebola patients. The drug was 100% effective in monkeys even after onset of fever, and was the one experts were most comfortable trying in people.
Cautioning that he was giving only his own opinion, Jahrling didn't sound very hopeful about using serum from recovered Ebola patients as a treatment, an approach suggested by the World Health Organization (WHO). He said seven patients in a 1995 outbreak in Kikwit, Democratic Republic of the Congo, received convalescent serum, but "it was impossible to tell if it had anything to do with their recovery."
Convalescent serum "hardly ever works in monkeys," although it can have an effect if it is highly concentrated, he added. Media reports today noted that Nina Pham, the Ebola-infected nurse in Texas, received convalescent serum from Kent Brantly, MD, who recovered from the disease in August.
Pinning hopes on a vaccine
Osterholm described an effective Ebola vaccine as the best hope for stopping an epidemic that has confounded all expectations and is likely to continue to do so.
He warned the audience to expect the unexpected as the epidemic continues: "Do not expect that anything carved in stone today won't be blown up by some stick of dynamite."
He also urged them to get comfortable with uncertainty and be honest with the public about it. "We're making this up as we go. We have to be mindful that we're making it up," he said. "One of the worst enemies we can have today is dogma."
As an example of current dogma, he cited the view that Ebola virus cannot spread through the air. Many virologists, he said, are concerned about the possibility of airborne transmission, and certain Ebola species have been known to go airborne in primates.
Reiterating points he has made in recent media commentaries, Osterholm said that men and boys in the affected West African countries come home every summer to harvest crops, and in October many leave to take temporary jobs elsewhere—a migration that increases the threat of spreading Ebola to neighboring countries.
"If West Africa was a can of gas waiting for a match to hit it, the rest of Africa is a tanker truck waiting for a match to hit it," he said.
He said that with efforts to smother the virus in Guinea, Liberia, and Sierra Leone faltering, a vaccine is "the only hope we really have."
"I do believe we can have an effective Ebola vaccine," he said, but the road to actually delivering a vaccine in quantity in African is long and full of obstacles. He commented that the reported US investment of $57 million in Ebola vaccine development is "a drop in the bucket."
Osterholm announced that he and Farrar of the Wellcome Trust will co-chair an "Ebola Vaccine Team B" with the goal of "laying out the challenges that must be met to get us a successful vaccine."
In a message to CIDRAP staff members, Osterholm said the team will include top global experts in vaccine research, development, manufacturing, distribution, and financial support.
In other comments, Osterholm said an international Ebola research agenda is urgently needed to answer a number of questions. For example, more virus isolates are needed for genetic studies, and information on clinical virology is sorely lacking.
Doubts about numbers
In another presentation, Joshua Epstein, PhD, a professor of emergency medicine in the Johns Hopkins School of Medicine, offered a mix of good and bad news concerning the projected spread of Ebola in West Africa.
He said he has reasons to doubt the recent worst-case estimate from the Centers for Disease Control and Prevention (CDC), which suggests that Ebola cases in the three countries could reach 1.4 million by January. That number assumes that there are 2.5 unreported cases for every reported case.
The CDC model that produced the number "is commendably explicit," he said, but "it heats up too fast and cools down too fast, in my view."
He said a standard model that considers behavior changes caused by the epidemic has a lower peak, but, unlike the CDC projection, doesn't "fade to zero." He suggested that the prevalence of disease will spur improvements in hygiene, burial practices, and use of protective equipment, adding, "I think we're seeing signs of this in Lofa, Liberia."
On the other hand, Epstein said he doesn't think Ebola will vanish from the region after the epidemic finally subsides. It's likely to be sustained, he predicted, by factors such as its persistence in animals, evolution, and human behavioral cycles of vigilance and complacency.
Epstein suggested that a way to replenish the decimated healthcare workforce in West Africa is to enlist Ebola survivors. "As far as we know, people who survive are permanently immune, so let's train them and use them as a workforce to control the epidemic. . . . They know the culture, they speak the language, they're trusted by others in their community."
In other comments, he said one thing anyone can do to help fight Ebola is to get a flu shot: "That will cut the background noise of symptoms and help clinicians be more efficient in their treatment of Ebola cases. It will help amplify the Ebola signal-to-noise ratio. So prevent the sniffle and get the flu shot."
See also:
Johns Hopkins Ebola forum page