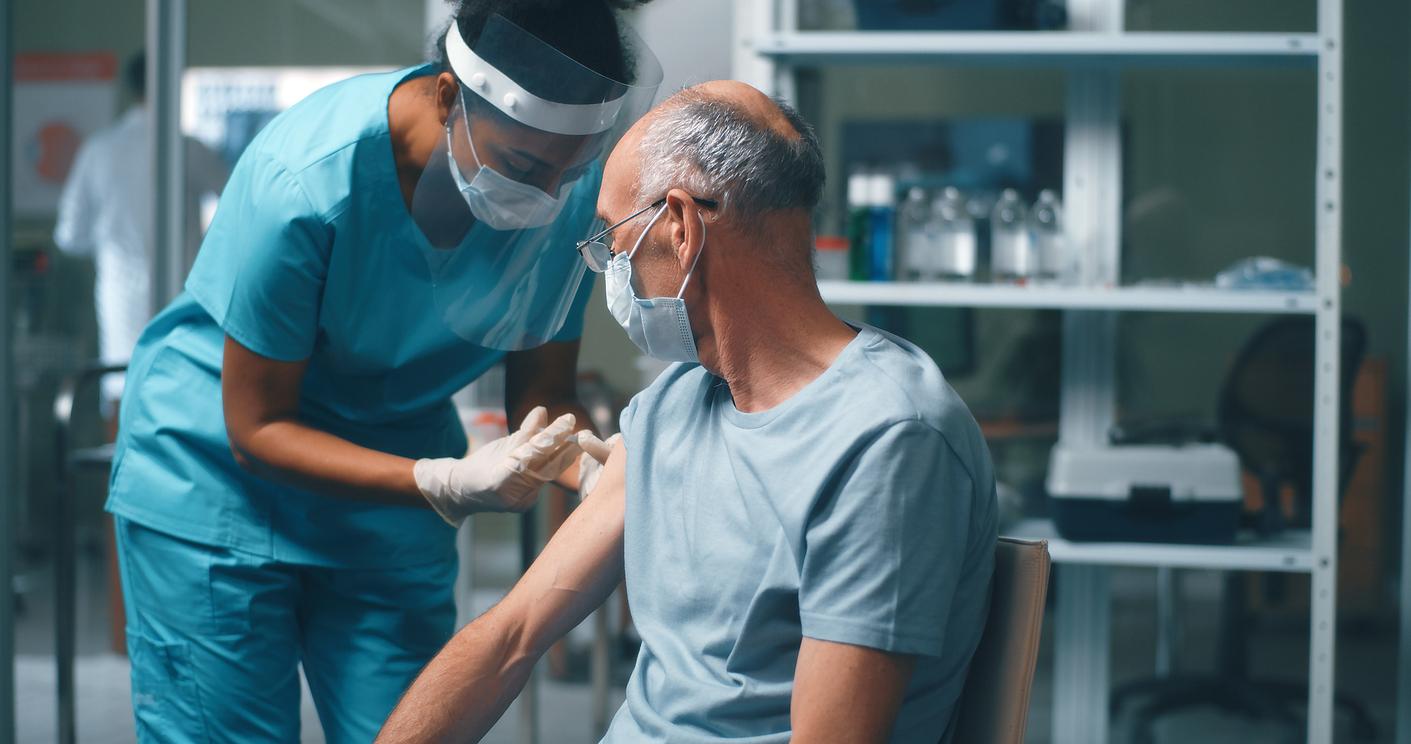
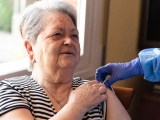
The Center for Disease Control and Prevention (CDC) vaccine advisory group today updated its advice for use of the respiratory syncytial virus (RSV) vaccine in adults, which now recommends that all people ages 75 and older receive a single lifetime dose, and that people ages 60 to 74 who have certain underlying conditions also receive a dose of the vaccine.
The Advisory Committee on Immunization Practices (ACIP) recommendations, which still need to be accepted by the CDC director, represent an evolving RSV vaccine landscape and availability, as well as new scientific findings that the group uses to guide it policy recommendations.
The group voted separately on recommendations for the two different age groups, and both passed unanimously on11 to 0 votes.
Evolving vaccine landscape and science
Since the CDC first recommended the vaccine for use in ages 60 and older in June 2023, the Food and Drug Administration (FDA) at the end of May approved a third RSV vaccine for use in older adults, an mRNA product made by Moderna. Earlier this month it approved expanded use of Arexvy, made by GSK, for at-risk adults ages 50 to 59.
The updated recommendations come ahead of the second respiratory virus season with the availability of an RSV vaccine for adults, which is thought to hospitalize 60,000 to 160,000 older adults each year, with the biggest burden in those who have chronic heart and lung problems and weakened immune systems.
ACIP has also grappled with the wording of recommendation. When it recommended the vaccine for use in adults ages 60 and older last summer, it did so with a "shared clinical decision making" recommendation, terminology the CDC uses when some uncertainty exists about benefits and risks.
However, the group has acknowledged that shared clinical decision making isn't simple in clinical practice and may contribute to unclear messaging regarding the need to vaccinate.
The group today postponed a recommendation for use of Arexvy in people ages 50 to 59, due to a lack of data that would support a population-based recommendation and lingering concerns about the risk of Guillain-Barre syndrome (GBS) following RSV vaccination.
Scaled back recommendations for ages 60 to 75
ACIP members acknowledged that the recommendation for adults ages 60 to 75 was less permissive than last year's advice, which would likely mean those who aren't in risk groups would need to pay out of pocket for the RSV vaccine, which could cost as much as $400. Several said the group's advice typically settles over time when new vaccines enter the market. Matthew Daley, MD, ACIP member and senior investigator with the Institute for Health Research at Kaiser Permanente Colorado, said a shift in recommendations reflects the nature of uncertainty, as scientists gather and analyze more data.
Members also welcomed a list of specific underlying conditions that its RSV vaccine work group pulled together, which they said will help with making clinical decisions for patients ages 60 to 75. Conditions include lung disease, cardiovascular disease, moderate or severe immune compromise, diabetes with end-organ damage, severe obesity, neurologic or neuromuscular conditions, advanced chronic kidney disease, liver disorders, and hematologic disorders.
To add flexibility for doctors treating patients in this age group, the list includes an "other chronic medical conditions" category for when health providers believe other patients are at increased risk of RSV complications.
After the vote, Jamie Loehr, MD, ACIP member and owner of Cayuga Family Medicine in Ithaca, New York, emphasized that the small but real risk of GBS weighed heavily on the group's risk-benefit assessments in the two age groups, given that it is a severe neurological disease that can hospitalized patients for months.