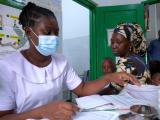
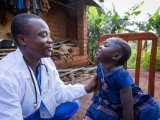
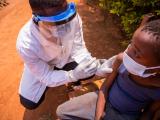
The final results of a phase 3 trial on the combination of a seasonal malaria vaccine and antimalarial drugs in children confirms a two-thirds decrease in the mosquito-borne illness, including severe cases and deaths, over 5 years in Burkina Faso and Mali.
Yesterday in The Lancet Infectious Diseases, a team led by researchers from the University of Science, Technology and Techniques of Bamako (USTTB) in Mali describe the comparison of seasonal malaria chemoprevention (SMC) with sulphadoxine-pyrimethamine and amodiaquine plus control vaccines, the RTS,S/AS01E vaccine plus placebo SMC, or SMC plus RTS,S/AS01E.
The World Health Organization (WHO) recommends monthly SMC during the rainy season, when malaria transmission peaks. GlaxoSmithKline's circumsporozoite protein-based RTS,S/AS01E vaccine is licensed for malarial control in Africa. Circumsporozoite protein is the antigenic target of the vaccine.
Superior to preventive drugs, vaccine alone
Children enrolled in the initial 3-year trial at ages 5 to 17 months in 2017 were randomly assigned to receive one of the three interventions until they were 5 years old. In the phase 3 trial, children randomly assigned to receive the RTS,S/AS01E vaccine were vaccinated at months 0, 1, and 2, followed by a booster dose at month 20.
Non-inferiority was defined as a 20% increase in clinical malaria in the RTS,S/AS01E-alone group compared with the SMC-alone group, and superiority was defined as a 12% difference in the incidence of clinical malaria between the combined and single-intervention groups.
In April 2020, 5,098 (94%) of the 5,433 children who completed the initial 3-year follow-up were re-enrolled in the 2-year extension study. The incidence of clinical malaria per 1,000 person-years at risk over 5 years was 313 in the SMC-alone group, 320 in the RTS,S/AS01E-alone group, and 133 in the combination group.
The combination of RTS,S/AS01E and SMC was superior to SMC (protective efficacy, 57.7%) and to RTS,S/AS01E (protective efficacy, 59.0%) in preventing malaria. RTS,S/AS01E was non-inferior to SMC (hazard ratio, 1.03). The protective efficacy of the combination compared with SMC over 5 years was comparable to that observed in the first 3 years (protective efficacy of the combination vs SMC, 57.7% and vs RTS/AS01E-alone, 59.0%).