Jan 19, 2012
NBSB names 6 new members
The National Biodefense Science Board (NBSB), created under the Pandemic and All-Hazards Preparedness Act of 2006, will have six new members to replace members whose 4-year terms expire Jan 31, the Department of Health and Human Services (HHS) said in a release yesterday. "NBSB members bring a broad range of perspectives and experiences, and over the past 4 years, the board has helped us improve federal policies and practices in disaster preparedness and response," Nicole Lurie, HHS assistant secretary for preparedness and response, said in the release. The NBSB was chartered in May 2007 and has provided recommendations to HHS on a variety of federal disaster preparedness and response issues, such as the research and development of medical countermeasures. New members are: John S. Bradley, MD, Rady Children's Hospital, San Diego; Nelson J. Chao, MD, MBA, Duke University; Emilio A. Emini, PhD, Pfizer, Inc.; Manohar R. Furtado, PhD, Life Technologies/Applied Biosystems; Steven E. Krug, MD, Children's Memorial Hospital, Chicago; and Sarah Y. Park, MD, Hawaii's state epidemiologist. In addition, John S. Parker, MD, retired Army major general, was nominated to continue on the board as chair. The new members replace Patricia Quinlisk, MD, MPH (outgoing chair); Ruth L. Berkelman, MD; Stephen V. Cantrill, MD; John D. Grabenstein, RPh, PhD; Thomas J. MacVittie, PhD; and Patrick J. Scannon, MD, PhD. Six additional members will remain on the board.
Jan 18 HHS press release
Hospital measles outbreak underscored need for isolation precautions
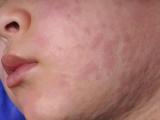
A measles outbreak at a Pennsylvania hospital in April 2009 sickened six patients, including a doctor who worked in the emergency department (ED), and triggered testing of hospital employees, according to a report today in Morbidity and Mortality Weekly Report (MMWR). After receiving reports of five individual cases, the Allegheny County Health Department determined that all patients had been in the same hospital ED. An investigation of serum samples of patients who had been treated there for rash and fever in the past several weeks identified a child who had arrived from India, who in follow-up testing was diagnosed as having measles. The patient had been in a room adjacent to the other patients, and the physician who became sick with measles had examined the boy. The physician had previously received three doses of a measles-containing vaccine. Since 2007 the hospital has required serologic documentation of measles immunity for new employees, but testing was inconsistent for previously hired workers. The facility conducted serology tests on workers who didn't have documentation. Of 168 employees, 72 (43%) did not have measles IgG titers on record and 8 (12%) did not have IgG protection. Those who weren't immune were vaccinated. No other employees got sick. In an editorial note that accompanied the report, the authors wrote that the outbreak illustrates the importance of isolation precautions for patients who have rash, cough, coryza, and fever and of serologic documentation for healthcare workers.
Jan 20 MMWR report
Study: Fake, substandard drugs threaten anti-malaria efforts in Africa
The battle against malaria in Africa is in danger because of poor-quality and fake anti-malaria drugs, according to a report in Malaria Journal. A team of international researchers examined anti-malarials collected in 11 African nations and in Asia en route to Africa from 2002 to 2010 and found that some counterfeits contained a mixture of wrong active ingredients, some of which may initially alleviate malaria symptoms but would not cure the disease and may even cause serious side effects. Some of the fakes also contained small amounts of artemisinin derivatives, possibly to enable them to pass authenticity tests. But at such low levels these drugs may make malaria parasites more resistant to artemisinin, the main malaria drug. Lead researcher Dr Paul Newton from the Wellcome Trust-Mahosot Hospital-Oxford University Tropical Medicine Research Collaboration in Laos, said in a Wellcome-Trust news release, "Public health organizations must take urgent, coordinated action to prevent the circulation of counterfeit and substandard medicines and improve the quality of the medicines that patients receive." The authors wrote that their study did not allow for an estimate of the prevalence of poor-quality drugs in Africa.
Jan 17 Wellcome-Trust news release
Dec 13 Malaria J abstract