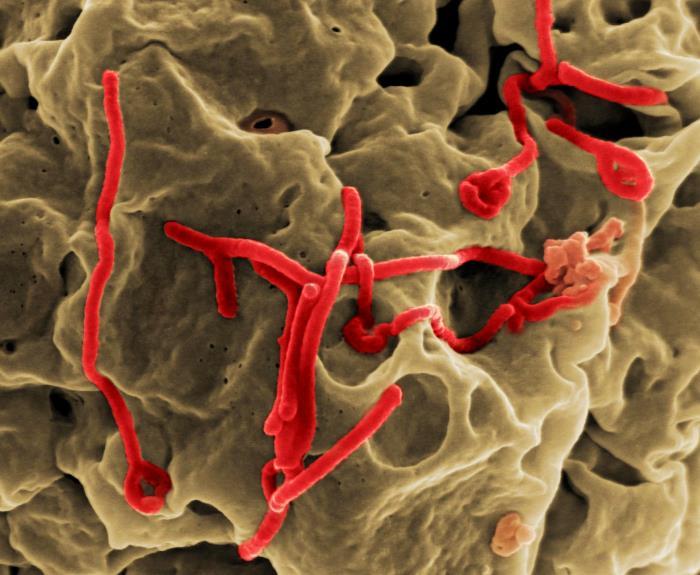
Doctors without Borders (MSF) today reported the first case of Ebola virus disease (EVD) in one of its own staff, and a media report said a health worker hospitalized with EVD in Atlanta is poised to receive convalescent serum from a British nurse who recently recovered from the illness.
As medical teams inside and outside West Africa continued to grapple with the disease, more nations announced support for the outbreak response, and a US House of Representatives committee heard from federal officials and others familiar with the medical and social challenges in battling the epidemic.
Health worker infection developments
In a statement today, MSF said the sick staff member is a woman from France who had been working in Monrovia, Liberia's capital. She was placed in isolation yesterday after coming down with a fever and tested positive for the virus the same day. The group said she will be evacuated to a specialized treatment center in France.
MSF has been at the forefront of the EVD response and has been working in the region since the outbreak began in March. The woman is the eighth sick foreign medical worker to be airlifted out of West Africa's outbreak area.
Brice de la Vigne, MSF director of operations, said in the statement that MSF applies strict protection protocols for its staff before, during, and after they spend time in the EVD outbreak countries. "This dramatically reduces the risk of transmission of the disease. However, the risk is part of such an intervention, and sadly our teams are not spared," he said.
It's unclear how the woman became infected, but MSF teams are investigating the contamination as part of standard procedures, the group said.
More than 2,000 MSF staff are working in the region, and the group is known to have meticulous infection control protocols.
In a related development, William Pooley, a British nurse who recovered from EVD and received the experimental drug ZMapp, recently traveled to Atlanta to give serum to an Ebola patient who was hospitalized in a specialized isolation unit at Emory University Hospital on Sep 9, The Independent, a newspaper based in London, reported today.
Pooley flew to Atlanta on Sep 12, with his trip paid for by the World Health Organization (WHO), according to the report. The patient has not been named but is thought to be an American doctor who got sick while working for the WHO in Sierra Leone. It's not clear if the patient has received the serum.
Last week, reports emerged that another medical worker who survived EVD, Kent Brantly, MD, who contracted the disease while working with Samaritan's Purse in Liberia, donated convalescent serum to Rick Sacra, MD, a clinician who got sick while serving with SIM in Liberia and is hospitalized at Nebraska Medical Center in Omaha.
A WHO expert group on Ebola therapies recently concluded that blood products from survivors may offer early help in the outbreak, while clinical trials of experimental drugs and vaccines proceed. Supplies of ZMapp have been depleted, forcing doctors to consider other treatment options.
Sacra's condition continues to improve, and though he tires easily, he is having long conversations with friends and colleagues, and the medical staff has brought in books and games to help him pass the time, according to a Sep 15 press release from the hospital. It added that doctors continue to be pleased with Sacra's progress.
Flow of donations continues
In response developments, the World Bank yesterday approved a $105 million grant to help pay for epidemic response activities underway in Guinea, Liberia, and Sierra Leone, according to a press release from the group. The funds are part of the $200 million in emergency Ebola mobilization funds first announced by the institution in early August.
The largest chunk of the grant money—$52 million—is earmarked for Liberia, the country with the most infections. The World Bank said Sierra Leone will receive $28 million and Guinea will get $25 million.
The money comes from a crisis fund intended to help poor countries recover from natural disasters or economic crises and will help finance hazard pay for people working in treatment and referral centers, medical care for exposed health workers, death benefits to families of health workers, and recruitment of medical staff from other countries. The World Bank said the fund will also help rebuild medical and public health systems.
Meanwhile, Australia said today it will donate an extra $6.4 million to help with the outbreak response, the Associated Press (AP) reported. In an earlier announcement the country said it had committed $903,000 to the fight against the disease.
Japan's government has donated emergency supplies worth $289,000 to Liberia, AllAfrica reported yesterday. The items include tents, sleeping pads, and other items.
At a US House Foreign Relations subcommittee hearing on Ebola today, a US Agency for International Development (USAID) official said one of the US aims in announcing its surge response to the outbreak yesterday was to inspire other countries to take similar steps.
Nancy Lindborg, USAID assistant ambassador, said there are indications that countries have more forward-leaning contributions in the works, including an announcement soon from the United Kingdom. "Over the next week, watch for the global response to ramp up," she said.
House committee seeks Ebola answers
For the second day in a row, federal health officials involved in the response appeared before a legislative committee. The House committee hearing covered similar ground to yesterday's Senate joint committee hearing, with legislators from both groups seeking answers about the safety of US troops to be deployed and how the multiagency response to the epidemic will be led.
Rep. Chris Smith, R-N.J., who chairs the House subcommittee on Africa, Global Health, Global Human Rights, and International Organizations, said, "Putting many more Americans in harm's way, we want no stone left unturned to protect them."
The panel learned that many of the US troops won't be involved with patient care, and that the military has good infection control procedures, complemented by a Centers for Disease Control and Prevention (CDC) Ebola training program that was developed with the help of MSF.
Smith and his colleagues raised concerns about a clinical hold that was placed this summer on Tekmira Pharmaceuticals' antiviral drug for treating EVD infections. The drug was developed with support from the Department of Defense, and the Food and Drug Administration (FDA) first cleared it for clinical trials in 2011, following promising findings in nonhuman primate studies.
The FDA placed a clinical hold on phase 1 studies in early July, and on Aug 7 modified it to a partial hold. In an Aug 7 press release the company said the action enabled potential use of TKM-Ebola in people infected with EVD. It said the hold relates to the multi-ascending dose portion of the phase 1 trial in healthy volunteers.
Luciana Borio, MD, who directs the FDA Office of Counterterrorism and Emerging Threats, said there are many reasons why a hold would be placed on clinical trials, but she wasn't at liberty to discuss the details of the Tekmira hold.
Borio said the FDA is working closely with the company to address the hold.
Kent Brantly, MD, appeared before Congress for the second day of hearings to describe the situation in Liberia. He said US health workers are being airlifted to the United States for treatment at a time when no medical gloves are available for purchase in Liberia's capital and the treatment center he worked at is turning away 30 patients a day.
"Military assets must be mobilized as quickly as possible," he urged, adding that he's pleased that the United States has committed to take the lead with the response. "It accurately represents the compassion of the American people."
Brantly repeated the appeal he made yesterday for outbreak responders to quickly arm home caregivers in the outbreak countries with materials to provide supportive care and protect themselves from EVD. He also said EVD survivors can be recruited as community Ebola educators: "It would help restore their dignity while tearing down the wall of stigma."
Three members of African medical groups also spoke to legislators, painting a stark picture of health infrastructures in the three countries that are worn out from recent civil wars and decades of neglect by the international health community.
Dougbeh Chris Nyan, MD, secretariat director for the Diaspora Liberian Emergency Response Task Force on the Ebola Crisis, said most hospitals in the country are still closed and lack medical supplies, and he hasn't seen a logistical plan to deliver promised materials to clinics.
Among several recommendations for the United States and its partners, Nyann called for Britain, France, and the United States to coordinate the response and for the countries to take control of and rebuild health systems in the region. He also urged global health officials to make use of private groups such as Catholic Relief Services to assist with the Ebola fight.
See also:
Sep 17 MSF press release
Sep 17 Independent story
Sep 15 Nebraska Medical Center press release
Sep 16 World Bank press release
Sep 17 AP story on Australian donation
Sep 17 House Foreign Relations subcommittee hearing materials
Sep 16 AllAfrica story
Aug 7 Tekmira press release