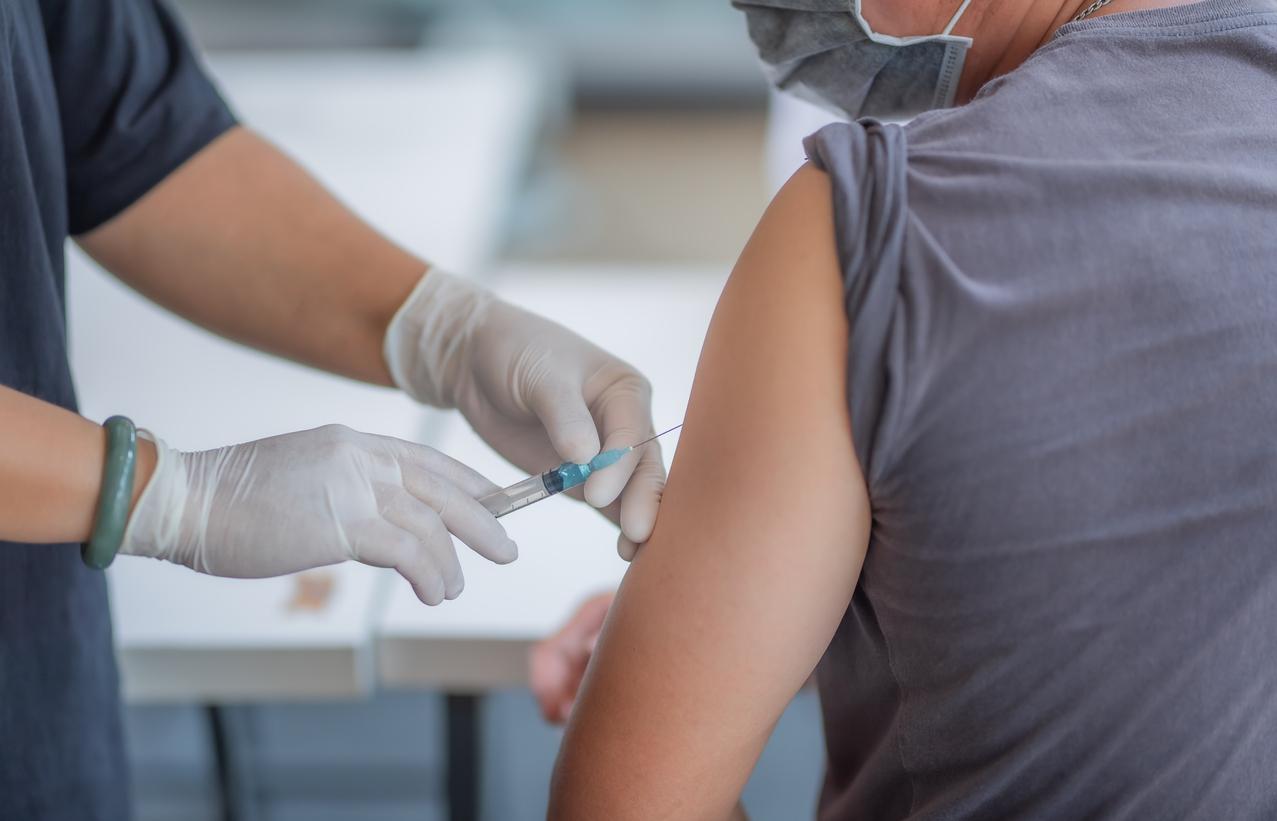
Today marks the start of the phase 3 trial of Moderna's COVID-19 vaccine candidate. The COVE trial (Coronavirus Efficacy) will enroll 30,000 people at 89 sites across the country, with half the recipients receiving two shots of the vaccine 28 days apart, and the other half receiving placebos. The study is blinded.
According to Moderna, the primary endpoint will be the prevention of symptomatic COVID-19 disease, and secondary endpoints will include prevention of severe COVID-19 disease (as defined by the need for hospitalization) and prevention of infection by SARS-CoV-2, the virus that causes COVID-19, regardless of symptoms. The trial is being conducted in collaboration with the National Institute of Allergy and Infectious Diseases.
If Moderna's candidate is successful, it will mark the fastest vaccine development timeline in history. The Phase 1 study of mRNA-1273 was dosed on Mar 16, 63 days from the virus sequence selection.
Modern's mRNA vaccine is the first US vaccine to enter phase 3 trials, but at least three more are soon to follow: a vaccine made by Novavax, a candidate that's the product of a collaboration between the University of Oxford and AstraZeneca, and one by Johnson & Johnson. Each trial will require 30,000 healthy participants, and all are part of the U.S. government's Operation Warp Speed, which has set the goal of producing, manufacturing, and distributing an effective vaccine against the novel coronavirus by 2021.
Globally, there are currently 27 COVID-19 vaccines in human trials.
Yesterday, Moderna announced it would receive an additional $472 million from the Biomedical Advanced Research and Development Authority (BARDA) to fund the late-stage trial of its messenger RNA vaccine. Initially, Moderna received $483 million from BARDA to support the scale up of vaccine production and clinical development.
"We thank BARDA for this continued commitment to mRNA-1273, our vaccine candidate against COVID-19," said Stephane Bancel, Moderna's Chief Executive Officer, in a press release. "Encouraged by the Phase 1 data, we believe that our mRNA vaccine may aid in addressing the COVID-19 pandemic and preventing future outbreaks."
Trump national security adviser COVID-19 positive
The highest ranking member of the Trump administration, national security adviser Robert O’Brien, has tested positive for COVID-19.
"He has mild symptoms and has been self-isolating and working from a secure location off site," the White House said in a statement. "There is no risk of exposure to the President or the Vice President. The work of the National Security Council continues uninterrupted."
In other government news today, 4,000 federal employees have tested positive for COVID-19, and at least 60 have died, according to a new report by the Labor Department. The employees are seeking worker's compensation and have filed claims asserting they contracted the virus while at work.
Oklahoma, Missouri, North Dakota set record highs
According to The Washington Post, Oklahoma, Missouri, and North Dakota all set record daily case highs today. Oklahoma's case counts have risen by 20% over the last few days. Oklahoma reported 1,401 cases today.
In total, the US has 4,271,095 cases and 147,253 deaths, according to Johns Hopkins University.
In Kentucky, Governor Andy Beshear is expected to announce today new restrictions to curb the outbreak in his state. Yesterday, White House coronavirus task leader Deborah Birx, MD, reportedly told the governor he needed to shut down bars as infection rates and test positivity rates were increasing.
Beshear ordered a statewide mask mandate over the weekend, and prohibited social gatherings of more than 10 people.
Other US developments
- A church revival in Alabama held last week has so far resulted in 40 cases of COVID-19. The Baptist church was in the middle of hosting a weeklong revival when two members said they tested positive.
- Thirteen players on the Miami Marlins have tested positive for COVID-19, after the first weekend of Major League Baseball games. The team has postponed at least two games.
- Today Senate Republicans unveiled their $1 trillion stimulus packages, which would see weekly COVID-related unemployment payments reduced from $600 to $200. The package includes $105 billion for schools, and a second direct payment for families and individuals.