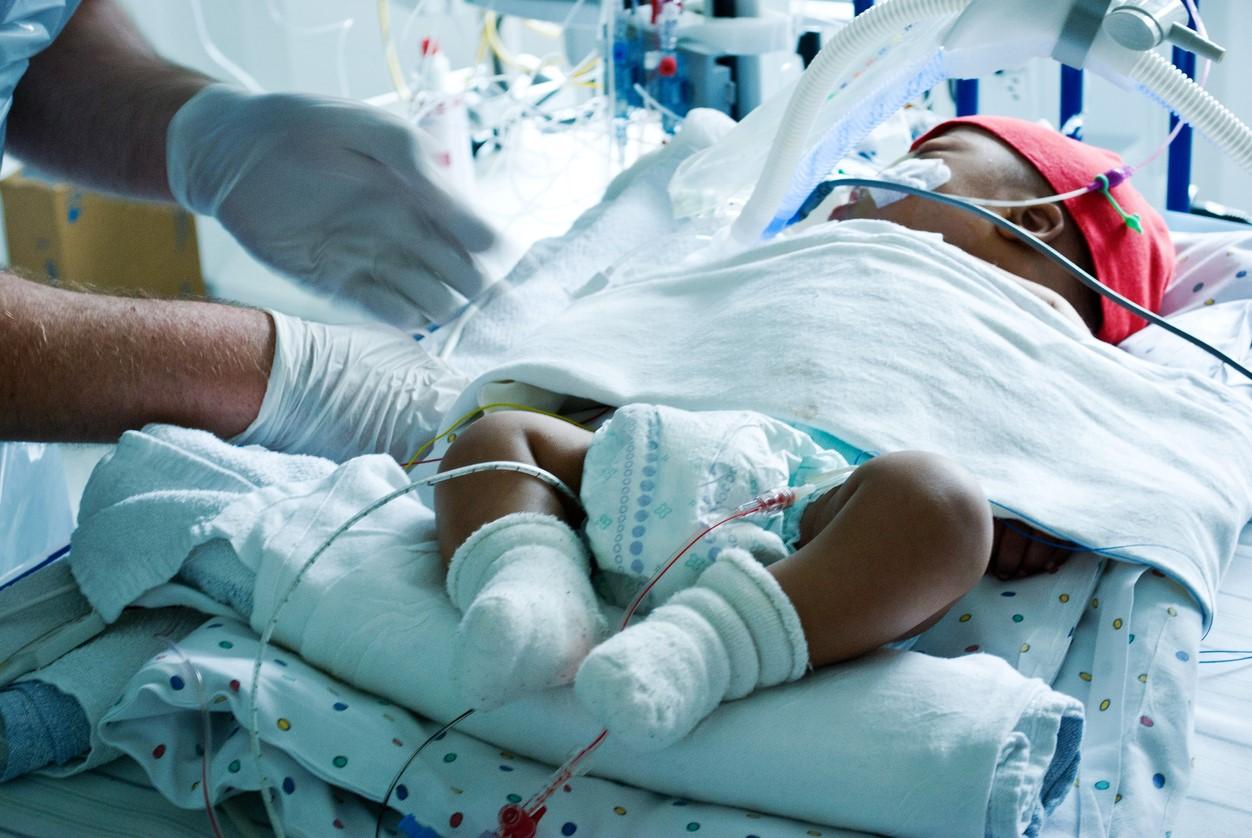
Over 81% of infants admitted to an intensive care unit (ICU) for respiratory syncytial virus (RSV) during the 2022 seasonal peak had no underlying medical conditions and were born full-term, finds a study published today in JAMA Network Open.
The Vanderbilt University–led surveillance study was conducted among 600 infants admitted to an ICU or other high-acuity unit for RSV at 39 hospitals in 27 states from October 17 to December 16, 2022.
The researchers enrolled 15 to 20 consecutively admitted infants at each site. The median patient age was 2.6 months, 60.2% were boys, 28.9% were born prematurely, and 81.2% had no underlying conditions.
They noted that RSV is the leading cause of respiratory disease–related hospital admissions in young children around the world.
"Although most children hospitalized with RSV are previously healthy and born at term, children with a history of prematurity or certain underlying medical conditions such as congenital heart disease, neurologic or neurodevelopmental disorders, chronic lung disease, and immunocompromising conditions are at higher risk for life-threatening RSV disease," they wrote.
Younger, premature infants most at risk for intubation
The primary indications for ICU admission were lower respiratory tract infection (LRTI; 99.0%) and apnea (temporary cessation of breathing) and slow heartbeat (12.8%). About a fourth (23.8%) of those receiving ICU treatment required invasive mechanical ventilation for a median of 6.0 days.
The highest level of respiratory support for nonintubated infants was high-flow nasal cannula (40.5%), followed by bilevel positive airway pressure (25.0%) and continuous positive airway pressure (8.7%). Infants younger than 3 months, those born before 37 weeks' gestation, and those with public health insurance were at elevated risk for intubation.
Intubated infants had a higher frequency of apnea, seizures, and increased sleepiness and a lower incidence of fever. Older infants had a higher frequency of fever, wheezing, rapid or shallow breathing, conjunctivitis, vomiting, and diarrhea, and younger infants had a higher incidence of apnea.
Identifying which infants are at risk for severe RSV disease is essential for assessing future clinical effectiveness and guiding product usage recommendations.
A higher proportion of intubated infants were in the Midwest (37.1%) and South (34.3%). Four infants received extracorporeal membrane oxygenation (ECMO), and two died, one of whom was receiving ECMO. The median duration of hospitalization for survivors was 5 days.
"Although mortality was rare, our findings emphasize the significant illness caused by RSV in young infants," lead author Natasha Halasa, MD, MPH, said in a Vanderbilt press release.
New monoclonal antibody can prevent RSV disease
For 15 of the 17 infants born before 29 weeks' gestation, there was no documentation of receiving the RSV monoclonal antibody (mAb) palivizumab—despite being eligible. Palivizumab is an intramuscular injection given monthly to high-risk infants during RSV season to prevent LRTI, but its cost is a significant barrier to global use.
The Food and Drug Administration (FDA) recently approved nirsevimab, a longer-acting mAb that neutralizes RSV and is considering a maternal vaccine to prevent RSV. "These products may protect both high-risk and healthy infants from medically attended RSV-associated LRTI," the researchers wrote. "Identifying which infants are at risk for severe RSV disease is essential for assessing future clinical effectiveness and guiding product usage recommendations."
Prevention of severe RSV may lower the risk of recurrent wheezing, asthma, or premature death as an adult. "The clinical severity information in this investigation can also help guide the design of upcoming RSV prophylactic and maternal RSV vaccine effectiveness studies," they concluded.