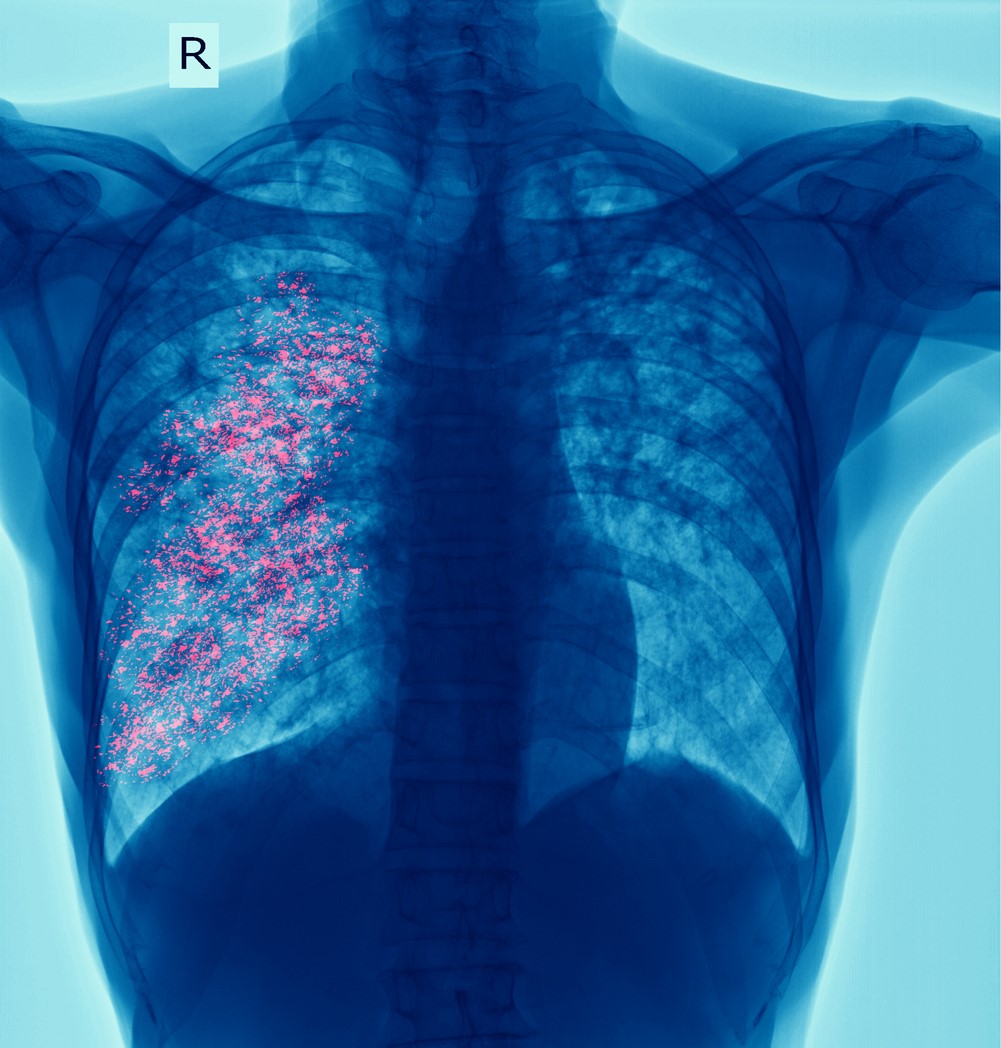
 The Bill & Melinda Gates Foundation and UK-based charitable organization Wellcome announced yesterday that they will provide a combined $550 million in funding for a phase 3 trial of a vaccine to prevent tuberculosis (TB).
The Bill & Melinda Gates Foundation and UK-based charitable organization Wellcome announced yesterday that they will provide a combined $550 million in funding for a phase 3 trial of a vaccine to prevent tuberculosis (TB).
The phase 3 trial will assess the efficacy of the candidate vaccine—M72/ASO1E (M72)—at preventing progression from latent TB to pulmonary TB. Trial investigators aim to enroll approximately 26,000 people, including people living with HIV and without TB infection, at more than 50 trial sites in Africa and Southeast Asia.
Developed by GSK in conjunction with the Gates Medical Research Institute, the vaccine combines the M72 recombinant fusion protein, which is derived from two Mycobacterium tuberculosis antigens, with GSK's proprietary adjuvant ASO1E. In a phase 2b trial, M72 showed roughly 50% efficacy in reducing pulmonary TB in adults with latent TB.
M72 would be the first licensed TB vaccine since the bacille Calmette-Guerin (BCG) vaccine, which was developed more than 100 years ago and is effective at preventing TB in children under 5 but ineffective in adolescents and adults. It's one of 17 TB vaccines in development.
TB remains one of the world's deadliest infectious diseases.
According to the most recent data from the World Health Organization (WHO), 10.6 million people fell ill with TB in 2021 and 1.6 million died. A WHO report in December 2022 estimated that a vaccine that was at least 50% effective in preventing TB in adults and adolescents could prevent up to 76 million cases and 8.5 million deaths over 25 years, avert as many as 42 million courses of antibiotic treatment, and prevent more than $41 billion in TB-related catastrophic household costs.
"TB remains one of the world's deadliest infectious diseases," Julia Gillard, chair of the Board of Governors at Wellcome, said in a Gates Foundation press release. "The development of an affordable, accessible vaccine for adults and adolescents would be game-changing in turning the tide against TB."
Additional details on the trial design and participants will be announced in the coming months.
 At the peak of the SARS-CoV-2 Omicron variant wave in Ontario, Canada, wastewater sampling conducted before the surge suggested that COVID-19 cases were 19 times higher than reported because of changes in clinical testing, according to a
At the peak of the SARS-CoV-2 Omicron variant wave in Ontario, Canada, wastewater sampling conducted before the surge suggested that COVID-19 cases were 19 times higher than reported because of changes in clinical testing, according to a 

 A single-center study in Canada found a low incidence of bacterial infections in infants with viral infections, including SARS-CoV-2, researchers reported today in JAMA Network Open.
A single-center study in Canada found a low incidence of bacterial infections in infants with viral infections, including SARS-CoV-2, researchers reported today in JAMA Network Open.













